If you have talked with your plant chemist or chemical provider lately, you are probably aware that prices for many of the chemicals used by generating plants have steadily risen over the past three years (see table). For power plants with evaporative cooling towers, cooling water chemicals have always taken the lion’s share of a typical chemical budget. Boiler and feedwater chemicals are used in very small quantities by comparison.
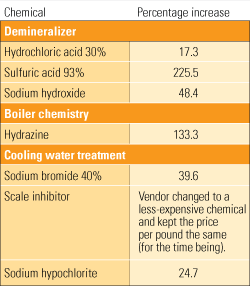
One utility’s chemical bill. Chemical costs rose sharply between 2005 to 2008. Source: M&M Engineering Associates Inc.
The cost increases are mainly caused by growing overseas industrial production. Other cost increases can be attributed to more transient conditions, such as the recently completed Olympic Games, during which steel and other heavy industry plants were reportedly shut down to improve air quality in and around Beijing.
By the numbers
Sulfuric acid is one chemical whose cost has increased significantly in recent years. Many plants use concentrated sulfuric acid to maintain pH in their cooling water and for regenerating the demineralizer. Sulfuric acid costs jumped 225% since 2005 for one utility, and its experience is almost certainly shared by many others.
Just as with air emissions, domestic production of sulfuric acid in the U.S. dropped precipitously since the early 1990s, while China’s has been increasing. Much of the world’s acid production is closely linked with steel manufacturing. Iron ore smelting is a major source of the sulfur dioxide used to produce sulfuric acid. Sulfuric acid can also be produced by the direct combustion of elemental sulfur, although that process is more expensive.
Concentrated sulfuric acid is used to process phosphate rock into phosphoric acid, which is used to produce fertilizer. Farmers worldwide are dependent on phosphate fertilizers to increase crop yields. It is estimated that 60% of global sulfuric acid production is used for producing phosphate fertilizers. Other major industrial users of sulfuric acid include the refining industry and manufacturers of nylon.
China, which is the second-largest exporter of phosphate rock, recently moved to increase the tariff on phosphate exports, forcing producers to keep more of the phosphate for local use. As a consequence, more of the sulfuric acid production also will be kept for domestic use. Considering the increasing acreage under cultivation for food and fuel in China and worldwide, it is unlikely that the price of sulfuric acid will be coming back down in the near future.
Chemical dependency
In addition to sulfuric acid prices, the prices of chemicals we depend on to prevent calcium carbonate scaling in cooling water and reverse osmosis systems have jumped significantly–and some chemicals have become more difficult to obtain at any price. Many of these anti-scaling chemicals are classified as phosphonates, and, as the name implies, they contain phosphorous.
Three common phosphonates used in cooling tower applications are AMP, HEDP, and PBTC. Though the complete mechanism is not understood, we know that these chemicals disrupt the crystalline structure of calcium carbonate, making it difficult for crystals to grow larger, attach to a surface, and accumulate to the point where they affect heat transfer in the condenser or create visible scale. Using these chemicals allows cooling towers to operate without scaling at higher cycles of concentration than would otherwise be possible.
These chemicals are typically made from yellow phosphorous, which comes from different sources than phosphate rock. However, pricing and demand can push the use of yellow phosphorous away from the production of complex organic phosphonate molecules used in cooling water treatment and toward fertilizer production. Additionally, many of the pesticides applied to crops are organo-phosphorous compounds, which also are manufactured from yellow phosphorous.
Recently, China placed a special tariff of 120% on the export of yellow phosphorous. This cost increase, coupled with increased demand, has caused the price of yellow phosphorous to rise sharply on world markets. In the past year, prices on common cooling tower phosphonates doubled or, in some cases, tripled.
Molybdate (molybdenum) is typically used in closed cooling water applications, though it is also useful at low levels in some open cooling water systems. Metal industry forecasts predict that molybdenum supply deficits are expected to be larger over the next two years. That will surely push already high prices for molybdate even higher in the future.
In sum, you should expect the price of common cooling water treatment chemicals to increase sharply as chemical costs are passed through the manufacturers of specialty chemicals and on to consumers.
What not to do
It is important to remember that cooling water systems rarely fail due to general corrosion–it would require close to 25 years of 10 mils per year of general corrosion (an unacceptably high rate) to cause a standard 1/4-inch piece of carbon steel piping to fail. Far and away the majority of the piping and heat exchanger tube failures in open or closed cooling water systems are caused by localized pitting. And localized pitting is the result of under-deposit corrosion.
Deposits are caused by a number of factors, including accumulations of silt and debris, calcium carbonate precipitation, and the growth of biofilms. Whatever the cause, the net result is the same: The area underneath the deposit becomes the anode and the area outside the deposit becomes the cathode; once the anode and cathode are established, corrosion is off to the races. Because it can be so localized (under a deposit), actual pitting or the potential for pitting in a specific area can be difficult to accurately access.
In rapidly fluctuating commodity markets as we have today, some might be tempted to cut back on treatment levels, to run closer to a minimum residual instead of in a safer range, or to increase the pH of the cooling water to reduce sulfuric acid costs. It also may be tempting to reduce biocide treatments to save money. Don’t fool yourself–these actions can have huge consequences on heat transfer, turbine backpressure, and corrosion in the cooling system that will far outpace the increased cost of chemicals.
Cutting back on treatment or applying treatment levels inconsistently will eventually lead to catastrophic failures that cost more to repair than any treatment regimen, regardless of rising chemical costs. Nevertheless, there are some things that you can do to ensure you are using the right chemicals to keep your system clean and reliable. The first step is to assess your current treatment practices and look for some better (and perhaps even less expensive) alternatives.
Below are two examples of lessons learned the hard way by a plant owner.
Case study #1: Pay me now or pay me later. It was a jump in the cost of molybdenum that contributed to problems in the mild steel piping in a chilled water system. As the cost of the metal increased and treatment costs kept pace, the decision was made to cut expenses by using much less treatment than is typical in these systems. The result was massive corrosion (Figure 1) that required replacement of major sections of the chilled water piping.

1. Out of balance. Failure to maintain proper levels of treatment resulted in a short-term gain and long-term loss for this piping system. Courtesy: M&M Engineering Associates Inc.
Once corrosion cells are established, they are very difficult to stop even if treatment is reestablished at the proper level later on. Corrosion cells also set up excellent homes for metal-munching bacteria. It also takes time for the symptoms to become obvious, so things may appear to be going great for a while, but sooner or later, the corrosion comes home to roost.
Case study #2: Microscopic real estate boom. If you fail to maintain either an effective biocide treatment regime or prevent scaling, then you should expect failures. Remember that microbiological fouling and inorganic deposits go hand in hand. Bacteria are looking for some real estate where they can settle down and multiply. Whether they start out as silt and debris or conventional calcium carbonate scale, deposits on cooling water piping look like brand new condos to acid-producing and sulfate-reducing bacteria. And once they move in, they are very difficult to evict.
The inorganic deposits protect the bacteria from biocides by absorbing or reacting with the biocide first. Similarly, once bacteria become established on piping, they form a biofilm that consists of a substance that traps debris and helps deposits to form. The combination can corrode most metals–copper, carbon steel, and even stainless steel can succumb to the effects of bacteria.
Figure 2 shows copper nickel tubing from a heat exchanger that failed due to bacteria deposits. When the tube deposits (which included calcium carbonate and silt) were removed from the sample, there was an area of pure copper metal around the failure. Preferential attack of one component of an alloy is a characteristic of microbiological-influenced corrosion. DNA analysis of the deposit proved that there were significant populations of corrosion-causing bacteria in the deposit.

2. Surface blemish. This heat exchanger tube failed because the bacteria did their work well. Courtesy: M&M Engineering Associates Inc.
Three Cs of successful cooling water treatment
There are three principles that, if followed, ensure the success of any cooling water treatment. Conversely, ignore them and your treatment system is bound to fail, resulting in corrosion and, ultimately, metal failure.
Clean. Keeping metal surfaces clean is the most important of the three principles. It is practically impossible for any chemical treatment to overcome dirty cooling water. Sometimes the dirt in your cooling water is just that–airborne dust, flyash, or coal dust that gets entrained in the cooling water. Cooling towers, after all are pretty good air filters. These particles soak up and inactivate chemical treatments, settle in condenser tubes and in cooling tower basins, deposit on tower fill, and in general provide areas for deposits to form and bacteria to hide out and flourish. Other forms of “dirt” in your cooling water are the living kind: clumps of bacteria that may come in with the makeup water, grow in pretreatment equipment such as softeners and clarifiers, or slough off from portions of the tower. The third form of “dirt” includes corrosion products such as rust and precipitates, including calcium carbonate.
Minimizing the accumulation of airborne particulate matter may be as simple as better housekeeping in the area of the towers. Better control of ash piles or dust suppression on roads and the coal pile may also produce significant benefits (Figure 3).

3. Airborne contaminants. The close proximity of the ash-loading area has caused particulate accumulation on this tower support. Courtesy: M&M Engineering Associates Inc.
Many have benefited from the use of side stream filtration, such as gravity multimedia filters, to remove suspended materials from the cooling water. These filters can produce a drastic reduction in particulates in the cooling water, regardless of its source, with a commensurate reduction in biocide and chemical demand in the tower.
Correct. The correct cooling water treatment is one that keeps the cooling piping and equipment clean and free of corrosion. It is not necessarily the treatment that contains the least expensive chemicals or uses the least acid or biocide. The correctness of the treatment can be seen in periodic inspections of the equipment.
Nothing in water treatment remains constant except change. It is critical that you are using the correct chemical treatment for your system. Changes in water sources, additional cooling demand, or changes in operating practices can result in a need for a significant change in cooling water treatment. Pitting rates are very flow and temperature sensitive. While stagnant conditions will generally reduce the dissolved oxygen differential between what is inside and outside a deposit, they give bacteria, particularly anaerobic bacteria, just the right conditions to multiply. Systems that cycle, or alternate between stagnant and flowing conditions, can be the most difficult to treat.
Over the years, the general industry trend in cooling water treatment has been to allow the pH of the cooling water to increase. This practice is based on the assumption (disputed by some) that corrosion rates for steel decrease significantly as the pH of the water increases. The potential for calcium carbonate precipitation also increases at higher pH, but some recent developments in polymers have shown promise in controlling precipitation even at these higher pHs. Some treatments now claim that they do not require the addition of any acid for pH control. This sounds very tempting when acid costs are increasing.
It is important to remember that the effectiveness of bleach as a biocide drops dramatically as the pH of the cooling water increases. The savings in acid can be swamped by increased costs for additional biocides to control bacterial growth at the higher pH. Bromide is used in addition to bleach to treat these more alkaline cooling waters, and it can also be expensive. (See “Biofouling control options for cooling systems” in POWER, September 2007.)
There are also a number of non-oxidizing biocides that can be very effective, but they typically have very strict requirements for their proper application and often require special handling precautions. These chemicals are typically also very expensive to apply on a regular basis. Before changing one area of the treatment, it is important to consider the effect that doing so will have on the rest of the cooling water chemistry and to account for the costs and benefits to the whole system. It may turn out that, even at today’s price, the additional sulfuric acid required to achieve the lower-pH cooling water will produce the best results with the lowest overall chemical treatment costs.
Consistent. Corrosion thrives on disparities. Consistent application of any cooling water chemical treatments is critical to the treatment’s success. Once corrosion cells are established under deposits, whether they are inorganic deposits or microbiological slime layers, they are very difficult to remove. Deposits change the surface chemistry at the metal/water interface regardless of the bulk water chemistry. Pitting corrosion often cannot be stopped until the deposit (or biofilm) is completely removed.
Cooling water chemistry and the consistent concentration of corrosion inhibitors is critical to controlling corrosion. The ability of treatment chemicals to prevent calcium carbonate precipitation depends on the amount of calcium and pH in the cooling water and the amount of residual chemical. On-line and/or grab sample testing must be routinely performed to ensure that all these parameters are within limits. Because many of these polymers are difficult to analyze for, assumptions are made that one parameter included in the treatment, for example, orthophosphate, is a good indicator of the amount of treatment in the system. However, if there are variable amounts of phosphate in the makeup water, this assumption may not be valid. These presumptions need to be reviewed for a treatment to be effective.
Regular treatments with biocides are also important, whether a system is operating or not. Bleach can control biological activity, but it does not stop it completely. So biocides, adjusted for the season and local conditions, need to be consistently applied, even if that means running the circulating water pumps for an hour or two.
Go with the pros
There will always be legitimate differences of opinion, particularly among water treatment vendors, about what the correct treatment regimen is for a particular cooling water system. Some differences are legitimate and are based on a detailed understanding of a particular cooling water system. Obviously, the experience and training of the person making the recommendations should weigh heavily in your decision.
There will also always be “new and improved” treatment regimes. Research into cooling water treatment polymers is very active, and there are often real innovations that permit one water treatment company to present a chemical treatment system that truly is superior to yesterday’s technology. However, in some cases “new” just means a repackaging of treatment chemicals that have been around for years and is different just for the sake of being different.
It is important to remember that new is not always better. In our rush to get the latest and greatest, we sometimes throw away some tried and true producers. If you are not sure if you are getting the straight story, you may want to consult with an independent cooling water specialist for recommendations of generic chemicals that you can buy from any vendor.
Because conditions can change in any cooling water system, the service you get from your chosen chemical treatment vendor can be of more value than the chemicals themselves. Having the correct treatment also means having an experienced set of eyes regularly examine, test, and make recommendations for change, as needed.
—David G. Daniels (david_daniels@mmengineering.com) is a principal of M&M Engineering Associates and a contributing editor to POWER.