The quality of any job results from the combination of the tools used and the expertise to use them properly. Oxidation-reduction potential (ORP) is one tool that, when properly applied and interpreted, can provide critical information about corrosion in feedwater piping. The promise of ORP is great: It may soon join pH and conductivity monitors as part of the standard suite of water chemistry tools. While still relatively new to fossil-fired power plants, ORP probes have been around for many years and are a common measurement tool at nuclear plants. In that application, they are used to control the addition of hydrogen to minimize inter granular stress-corrosion cracking in boiling-water and pressurized-water reactors.
ORP probes also have been used to control the addition of oxidizing biocides and sulfites in cooling towers to dechlorinate water before it is fed into the makeup water reverse-osmosis (RO) system. How ever, the use of ORP to minimize feed water corrosion in ferrous and mixed metallurgy systems is by far the most complex and challenging application of this technology, but may also produce some of the largest benefits.
Parallels to pH
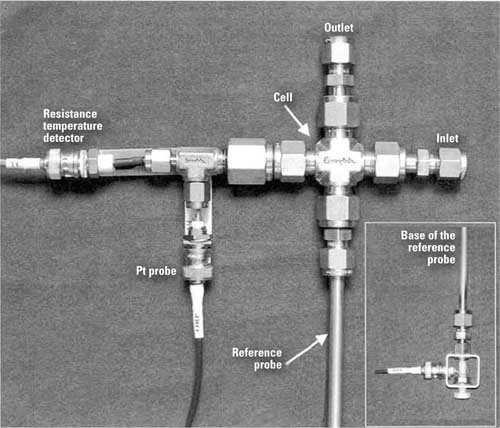
An ORP analyzer consists of a pair of electrodes—one for measurement and the other for a reference potential (Figure 1). The most common reference electrode is a standard silver/silver chloride (Ag/AgCl) wire in a potassium chloride solution, the same as the reference portion of a pH electrode. The measuring electrode consists of a small piece of inert metal, typically platinum. Under oxidizing conditions, the measuring probe loses electrons to the solution, which creates a positive potential; in a reducing environment, electrons are donated to the probe, producing a negative potential.

1. ORP measurements at fossil-fired plants may help control corrosion in feedwater systems. Source: POWER
There are other similarities between ORP and pH. Most important, perhaps, is that the purer the water, the more difficult it is to get a reliable response from the probe. In open systems-such as a cooling tower or raw-water feed to a reverse osmosis system, the measurement is fairly routine and stable. Measuring the ORP in feedwater requires careful control of many factors.
Control Oxidizing Biocides
For years, power plants have used oxidizing biocides to control the buildup of algae and other biofouling species in cooling water systems. In the past, chlorine gas (Cl2) was the most common biocide. Concerns about Cl2 safety, accidental release, and the effectiveness at alkaline pH levels has led many plants to switch to other oxidizing biocides- such as combinations of hypochlorite and bromide, chlorine dioxide, or chloro-bromo-hydantoin.
Regardless of the actual chemicals used, oxidizing biocides all have a common mechanism—they oxidize proteins in the cell membrane, which ensures the death of any cell the biocide touches. Economics, environmental concerns, and corrosion are all good reasons why plants would like to add just enough biocide to do the job.
For chlorine and chlorine-based biocides, the standard control point has been the residual chloride concentration found during treatment. This test can measure either the combined or free residual chloride. The assumption is that if sufficient combined or free chloride is in the water, the water must be toxic to biofouling species. However, studies have shown that neither combined nor free chloride is a reliable indicator of a biocide’s effectiveness at killing bacteria.
There are many factors that affect the ability of an oxidizing biocide, including pH, the chemical form of the chlorine or bromine, the effect of other oxidizing or reducing species in the water, and the biological demand of the water. For chlorine-based treatments, the combined residual chlorine test includes not only hypochlorous acid but also myriad chloramines and other chloro-organic compounds. Some of these compounds are very poor biocides and may even interfere with the active compounds.
While residual chlorine is not an effective predictor of controlling biological fouling, ORP is. Numerous studies have shown excellent correlation between the death rates of various viruses and ORP. For this reason, some facilities use an ORP measurement to control biocide feedrates.
The set point at which ORP stops the growth of biofouling species will vary for each cooling system, so it is best to do site-specific testing to determine ORP versus bacteria count guidelines. The ORP set point may need to be re-evaluated if there are major changes to the cooling-system water quality or metallurgy. In certain cases high background ORP may make control using this method difficult, but for many, the use of ORP, in con junction with chemical monitoring, can optimize biocide feedrate for changing water conditions.
Optimum Feedwater Chemistry
Pitting and under-deposit corrosion in the feedwater and boiler often begin when iron or copper react with O2. This O2 can be supplied by dissolved 02 in the feedwater or the oxygen that is part of the water (H2O) itself. Active corrosion has historically been associated with the most highly oxidized forms of the metal oxides. For example, hematite or iron (III) oxide tends to be porous and susceptible to the formation of under-deposit corrosion cells. Magnetite, the more reduced form of iron oxide (a combination of iron II and iron III oxides), forms a more tightly adhering and passivating layer on the metal surface.
For this reason, the focus has been to create a constant reducing atmosphere in feedwater and boiler piping to minimize corrosion and transport of corrosion products to the boiler. The primary method was mechanical O2 removal supplemented by the addition of a chemical scavenger. This combination virtually eliminates dissolved 02 in the feedwater during normal operation.
But simply removing 02 is not enough. The industry chose chemicals—such as hydrazine—that not only reacted with dissolved O2, but could also react with metal oxides—for example, converting hematite to magnetite. The widespread use of hydrazine and hydrazine substitutes at fossil-fired plants occurred, in part, because of their ability to passivate metal under certain conditions. Every O2 scavenger has a working temperature and pH range wherein it is most effective at scavenging 02. In addition, all organic-based or volatile O2 scavengers also break down into simpler organic molecules in the steam cycle.
The amount of O2 scavenger residual that power plants maintain in their feedwater has swung back and forth over the years. For a long time, the "more-isbetter" philosophy prevailed. For some, excessive use of O2 scavengers, particularly hydrazine, was thought to serve three purposes; reacting with dissolved O2, providing a more highly passivated system, and controlling pH as the excess hydrazine decomposed into ammonia. ORP measurements Jess than -300 mV can be generated under such conditions.
A few of the plants that were most vigilant in eliminating dissolved O2 suffered failures in feedwater piping from flow-accelerated corrosion (FAC). Subsequent research showed that when the dissolved O2 concentration in the feedwater was below 1 ppb or when the O2 scavenger residual was greater than 20 ppb, the corrosion rates attributed to FAC in all-ferrous units increased sharply.
The problem of FAC, combined with the introduction of oxygenated treatment to the U.S. by the Electric Power Research Institute in 1991, convinced many utilities to eliminate chemical O2 scavengers altogether. For plants with all-ferrous metallurgy in the feedwater portion of the plant, increasing the ORP reduced the potential for FAC, although not as much as full conversion to oxygenated treatment.
Mixed Metallurgy
Some background is necessary to understand why ORP holds such great promise for minimizing feedwater system corrosion.
Seeking the advantages of oxygenated treatment, a few utilities with copper-alloy condensers and/or feedwater heaters tried to follow suit and eliminate chemical O2 scavengers. The result was a significant increase in copper corrosion and corrosion-product transport problems. Once this was identified, hydrazine feed was quickly resumed.
The difficulty for mixed metallurgy units is adding sufficient O2 scavenger to prevent copper corrosion while minimizing iron corrosion and the potential for FAC in the ferrous-based components. One problem created by copper and copper oxide transport into the boiler is the potential for the copper to form a copper oxide compound that can be volatilized into the steam only to precipitate on the high-pressure turbine.
Operators of units with mixed metallurgy systems face a dilemma. If they operate in an oxidizing environment they experience copper corrosion; if they add sufficient scavenger to create a reducing environment, they run the risk of FAC. The latter problem is exacerbated by air in-leakage that can create high dissolved O2 and ammonium carbonate levels in the feedwater. This is further complicated because conditions are seldom stable in many of these units, because they load follow and/or cycle on and off daily. Changes in pH, dissolved O2, scavenger residual, ammonia concentration, deaerator performance, and feedwater flow rate all can change dramatically as load changes.
Plant operators can try to juggle controllable parameters (pH, dissolved O2, and scavenger residual) but this will not give a total picture. What is missing is a single measurement that could provide an indication of how all these conditions interact.
Is ORP that measurement? The answer is a qualified yes. Properly configured and interpreted, ORP does provide the plant staff with information on the condition of feedwater piping that, together with additional information, can minimize feedwater corrosion and corrosion product transport into the boiler and turbine.
High-Purity Waters
The problem of ORP in high-purity waters is two-fold—getting a reliable result and learning how to interpret that result for your facility.
The most accurate ORP measurement is taken in-situ or at temperature and pressure. Specialized probes are used at nuclear plants both in the reactor core and in secondary (non-radioactive) areas of the plant.
In a Swedish study, the ORP of a secondary feedwater circuit of a pressurized water reactor was monitored with probes installed in an "autoclave," or special sampling loop that allowed a side stream of feedwater to flow past pairs of ORP probes before being returned. The probes operated between 80 and 120 bar (1,200-1,800 psig) and temperatures as high as 450F. Under these conditions, the ORP probes responded very quickly to changes in dissolved O 2 and hydrazine concentration. Both the Ag/AgCl reference electrode and measurement probe had to be specially designed to withstand these temperatures and pressures. Similar in-situ ORP studies have been performed in Germany, Italy, and Spain.
The cost of developing a separate isokinetic sampling loop and high-temperature and -pressure probes may scare many fossil-fired plant operators away from using in-situ ORP except in the most unusual circumstances. But this should not steer fossil-fired plants away from ORP altogether. While not as accurate or responsive as an in-situ device, important information can be gathered by monitoring ORP on a conditioned sample if specific steps are also taken to minimize changes caused by sampling and to stabilize the readings.
A system’s ORP, and therefore its corrosion potential, is affected by several factors. Therefore, if any of these change significantly in the sample collection process, the corresponding ORP measurement will change also. The problem is not only that the response of the ORP probe on a conditioned sample may be different than the actual ORP in-situ, but that the sample line itself may change the ORP of the water. For example, a sudden increase in the ORP in the feedwater caused by a loss of hydrazine feed could be tempered by a long sample line and the reaction of the water sample with deposits in the line. The insulating nature of high-purity water creates the same challenges for ORP measurement as for pH. To ensure accurate ORP readings, the following guidelines are recommended:
- Eliminate streaming potentials by grounding the sample chamber and using stainless steel sample lines and sample chamber.
- Maintain a sample flow rate of at least 100 ml/min to 150 ml/min.
- Use a flowing reference electrode or a combination ORP electrode with an annular, diffusion reference junction to provide faster and more sensitive response. Some users have had stability problems with this type of probe and have gone back to a gel-type reference electrode in spite of the difference in response.
- Avoid gas-permeable tubing, such as Tygon, in any ORP sample line.
- Keep sample temperature relatively constant. There is no way to automatically compensate for changes in ORP caused by changes in temperature.
- Ensure that all fittings are tight and that any sample reservoirs contain no trapped air.
- Standardize and clean ORP probes and check their responsiveness regularly. An in-line sample filter may help prevent metal oxide films from accumulating on the probe.
Remember, ORP is not pH. There is no iso-electric point or theoretical slope as with pH. Differences in the probes themselves affect the magnitude of the response as well as whether the probe is reading a high-conductivity standard solution or the high purity of a feedwater sample. There are also plant-specific differences- such as feed water heater metallurgy and sample-line length and configuration.
While the ORP reading at your plant may not be the same as at another facility, you should be able to quickly determine what reading is normal under passivated conditions. This should alert you when the reading indicates that something in the system has changed.
—David G. Daniels (david_daniels@mmengineering.com) is a principal of M&M Engineering and a contributing editor to POWER. This article is derived from an article that appeared in the July/August 1999 issue of POWER.