By now, most in our industry are familiar with the explosion that occurred at the Kleen Energy plant in Middletown, Conn., in February 2010 (Figure 1). Personnel at this 620-MW combined cycle plant were performing a series of gas blows to clean the fuel lines prior to startup when a large mass of accumulated methane ignited. The blast killed six people, injured more than 50, and heavily damaged the nearly complete plant, which was scheduled to begin supplying power in June.
In addition to the loss of life and damage to the site, the explosion significantly disrupted the lives and livelihoods of many in the surrounding area, as state and federal authorities initially feared a terrorist attack and then took over the plant to conduct an extensive investigation into the cause of the blast. The Occupational Safety and Health Administration (OSHA) would ultimately hand down a total of $16.6 million in fines, the third-largest amount ever levied for a single incident. Criminal charges against the companies involved were considered (though ultimately none were brought), and several civil lawsuits remain pending.

1. The Kleen Energy plant in Middletown, Conn., was heavily damaged during a gas blow in 2010 after a mass of natural gas that had accumulated between the heat recovery steam generators exploded. Source: Chemical Safety Board.
The root cause of the blast was the use of natural gas as a cleaning agent during the gas blows. Though long a standard practice, similar (though less spectacular) accidents in the past had highlighted the risks if careful procedures were not followed. Many experts had begun to advocate the use of nonflammable media such as compressed air or nitrogen.
Among many other recommendations in the aftermath of the Kleen Energy explosion, the U.S. Chemical Safety Board’s (CSB’s) report concluded that the release of large amounts of natural gas in the vicinity of workers and potential ignition sources was “inherently unsafe.” It urged OSHA and other industry authorities to ban the use of flammable gases for cleaning and purging fuel gas piping systems. It also requested that turbine manufacturers develop guidelines for system cleaning using methods besides natural gas.
In August 2011, the National Fire Protection Association (NFPA) released provisional standard NFPA 56, which contained such a ban. The NFPA specifically noted that the CSB’s research found that many facilities have begun using air or nitrogen for blowing and cleaning processes because this is viewed as a safer practice than using natural gas.
Though only advisory, NFPA standards are widely viewed as persuasive to regulating agencies. The state of Connecticut has also enacted such a ban, though so far no other state has followed suit. In light of these developments, this article reviews some best practices for using air and nitrogen as cleaning agents.
The Basics of Fuel Line Cleaning by Gas Blows
To understand the issue it is necessary to understand the reasons and procedures behind cleaning natural gas piping.
Natural gas–fired combustion turbines are complex instruments designed to very tight tolerances, and consequently, they require a high degree of cleanliness. Contaminants in the fuel line can clog fuel combusters, cause premature system wear, and damage turbine blades. If a foreign object entering a turbine at operating speed is large enough, the damage can be catastrophic, though fuel lines are typically not of sufficient diameter to admit such debris. Even when no foreign matter reaches the turbine itself, dirty fuel lines can cause fuel flow and combustion problems, increasing stress on the system.
Manufacturer guidelines for system cleanliness dictate limits on particle size that are typically no larger than 20 µm and may be as small as 5 µm. Adhering to these limits requires a high level of attention to excluding foreign material.
Though the fuel supply lines for a gas-fired power plant may appear to be a sealed system to laymen, there are in fact many ways in which contaminants can be introduced.
The first, and perhaps most obvious, is during initial construction. When the fuel supply lines are being assembled, the system is typically open to the atmosphere, allowing the direct introduction of contaminants. Though the use of foreign material exclusion covers can reduce the potential for contamination, it cannot completely eliminate it. Anyone who has been involved in the maintenance of piping systems can relate stories of finding stray objects such as nuts and bolts, gaskets, tools, welding debris, and even extraneous items such as beer cans or snack wrappers inadvertently introduced by construction personnel. This author has even encountered a few situations where such debris was intentionally introduced by malicious individuals as an apparent form of amusement. Even when the human occupants of a construction site take due care not to introduce such debris, vermin such as rats and mice may do so while in search of a hiding or nesting place.
In addition to foreign material, as long as the piping is open to the air, it is subject to experiencing condensation inside the pipes, which can lead to rust and corrosion. This can occur even when exclusion covers are scrupulously used, as long as the environmental conditions are right.
Weld slag and spatter is another common source of contamination. This can be reduced by the use of careful welding processes, but it is virtually impossible to completely eliminate. In addition to weld slag, residual pipe mill scale (a by-product of the hot-rolling process) will eventually flake off the interior of the piping if not removed before assembly.
Most of these same concerns apply during system maintenance, especially when a fuel line must be opened to replace or repair a component, or during pigging.
Even if construction and maintenance procedures are successful in minimizing contaminants, there are other ways foreign material can enter a piping system. If a system has been hydrotested or cleaned with water jet flushing, there will be residual water and water vapor that must be removed, typically with compressed air blows or flushing with low-humidity air.
What Happens During a Gas Blow
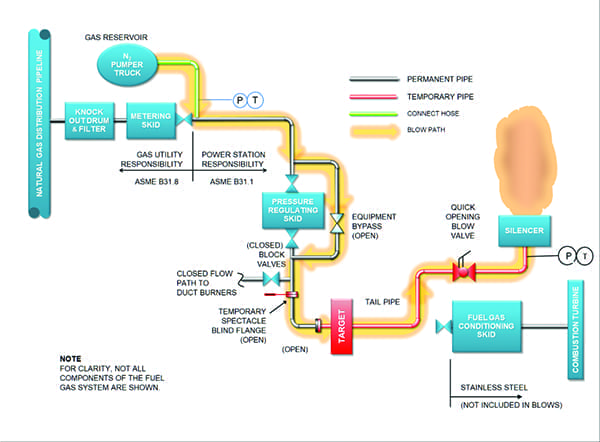
Conducting a gas blow is not simply a matter of blasting compressed gas through a piping system. Careful planning and calculations are necessary to ensure that sufficient, but not excessive, pressure levels are achieved as the slug of gas moves through the lines. Too little, and the desired cleansing will not take place; too much, and damage may occur. The geometry and characteristics of each segment of the system must be taken into account, as must the precise flow path for each blow. In-line elements that may interfere with the flow, or that cannot withstand the force of the blow, may have to be removed, and temporary piping may need to be put in place.
The outlet for each blow must be carefully planned for safety and system cleanliness. Depending on the location of the plant, additional planning may be necessary to reduce noise and environmental impact from the blow discharge. For obvious reasons, personnel must be excluded from the discharge area because of the potential for injury. Gas and contaminants exit the system at high velocity (Figure 2) and can carry dangerous projectiles with them, such as the aforementioned tools, bolts, weld slag, and vermin remains.

2. The first blasts from a gas blow can carry substantial amounts of debris and contaminants. Source: CSB
Once the system has been divided into appropriate segments for cleaning, the sequence of blows must be planned. Blowing usually follows the path of natural gas flow during operation. Typically, a blow will begin with larger-diameter piping, with the smaller-diameter lines isolated from the main line. Then, the smaller-diameter lines are cleaned, often by using the larger-diameter pipes as a gas reservoir. Care and planning is necessary to ensure that debris is not reintroduced into previously cleaned sections.
Once the plan is in place, each segment is cleaned using a series of high-pressure blows. Cleaning—both removal of contaminants from interior surfaces and ejection of loose debris from the piping—is typically accomplished with anywhere from 10 to 20 pulses to more than 100 pulses of gas. Blows are initiated either by fast-opening a valve from the pressure reservoir into the section to be cleaned or by inserting a rupture plate into the line that will burst at a specified pressure. The latter is more time-consuming, as a new rupture plate must be inserted for each blow, but it places less stress on the system, as repeated rapid cycling of valves can cause premature wear, especially on larger-diameter lines.
A blow is considered complete when the required cleanliness level is achieved. This is a somewhat subjective determination, but it is typically measured by placing a strike target (usually a sheet of metal or wood) in front of the outlet. Debris exiting with the blow will strike the target and leave a mark. The blow is considered finished when a certain minimum number of debris marks is achieved.
Natural Gas: Convenient but Risky
There are many inherent risks in conducting a gas blow, which involves discharging high-pressure gas through a piping system in a series of rapid bursts. These include added stress on valves, fittings, gaskets, and other connections such as sensors and gauges that may be attached to the fuel line. The main risk, however, is that the blow is vented into the open air (Figure 3).
3. When natural gas is used for a gas blow, care must be taken to ensure that the discharge does not accumulate. Conducting a blow like this one, vented into a partially enclosed space, is asking for trouble. Source: CSB
Natural gas has traditionally been used for cleaning because it is readily available on site using existing feed lines and does not require the system to be flushed afterwards. The above concerns exist regardless of the cleaning medium being used, but when using methane, there is the added risk that the blow creates a cloud of potentially explosive gas.
Consequently, when using natural gas for a blow, exhaustive efforts must be taken to remove potential sources of ignition: grounding the system to eliminate static electricity, deenegizing the site, and clearing the area of anything that could create sparks. Even with these efforts, several gas-blow explosions are believed to have been caused by nothing more than metallic debris exiting the piping and causing a spark when it struck a nearby object.
In addition, great care must be taken to ensure that the discharged gas does not accumulate at levels above the lower explosive limit (the concentration in air above which combustion can occur). This is especially true when the gas exits the system in partially confined areas. In the case of the Kleen Energy blast, gas was released between two heat recovery steam generators and allowed to build up until an explosion occurred. Although best practices dictate that the natural gas be vented only to open areas, such gas clouds can still build up unpredictably. Thus, the risk of an explosion can be reduced but never completely eliminated. It is for these reasons that the CSB and NFPA deemed the use of natural gas for line cleaning too dangerous for continued use, especially since several safer alternatives exist.
Leaving aside the safety issues, there is another reason to avoid using natural gas for cleaning. Methane is a potent greenhouse gas and is 21 times more efficient at trapping heat than CO2. Though the total amounts of methane released during a single cleaning project may not be great compared with the CO2 emissions from an operating plant over the long term, this is still a point worth considering, especially since more environmentally friendly alternatives are available.
Compressed Air: Safer, but Less Effective
One key thing to remember is that there is nothing about methane itself that cleanses the piping during a blow. Rather, it is the force of the gas blown through the line that removes debris and flushes it from the system. Thus, other gases can be employed using the same procedures. Compressed air has the advantages of being nontoxic and essentially free. Typically, air blows are conducted by attaching high-pressure air compressors to the system. To ensure sufficient volume for the blow, the air may be stored in a receiver (storage tank) after being compressed.
One disadvantage is that air drawn from the atmosphere contains water vapor, and this vapor will be converted to liquid water upon compression. Unless it is removed before the blow, that water will be introduced to the system. In humid climates, this is a significant concern. Thus, after compression, the air is typically passed through a dryer. Dryers can be either refrigerant-based (similar to a common dehumidifier) or desiccant-based. The latter, which pass the air through a hydrophilic material such as activated alumina or silica gel, are significantly more effective.
The main limitation of using compressed air is that there is typically insufficient storage space for the air upstream of the section to be cleaned as compared with other methods. Consequently, more and smaller blows must be conducted, increasing the expense and time required for the operation.
Nitrogen: Effective, but More Demanding
Nitrogen has some notable advantages over compressed air. It is cleaner and contains no water vapor that must be removed. More significantly, because it is stored as a liquid and delivered on site in mobile pumper trucks (Figure 4), it can deliver much greater capacities and pressures than a compressed air system. This makes for a faster, more efficient overall process. Nitrogen is more costly than compressed air, but the total cost is still comparable to a blow conducted using natural gas.

4. Liquid nitrogen is readily available in large quantities for many industrial uses and is typically delivered in mobile pumper trucks. Courtesy: Jerry Energy Service Co., Ltd.
It is important to remember that though nitrogen is classed as an inert gas, inert does not mean safe. Though it is not explosive like methane, nitrogen carries its own set of risks, the biggest of which is asphyxiation. Even though air is 78% nitrogen, nitrogen is not safe to breath alone—a few breaths of pure nitrogen can be deadly. A 2003 study by the CSB found that an average of eight people a year died from nitrogen asphyxiation between 1992 and 2002.
When free nitrogen is released into an area, it tends to displace oxygen. Nitrogen has no odor, so without specialized monitors, the first sign of an oxygen-deficient atmosphere may be unconsciousness. Worse, as the oxygen level drops, the brain’s ability to make good decisions falls with it, so an individual in such an environment may be unable to perceive what is happening and escape to safety.
Atmospheric monitoring devices must be in place any time pure nitrogen is being used. Interior spaces where nitrogen might escape must be properly ventilated, and rescue procedures must be prepared in case personnel are overcome by exposure to a nitrogen cloud.
Other Methods
In some circumstances, other cleaning methods may be appropriate. Water jet flushing, or hydrolazing, involves feeding a special nozzle and hose through the piping system in order to blast the interior with very high-pressure (10,000 psig and up) water to remove contaminants. This is a costly, labor-intensive process and also produces wastewater that must be cleaned before disposal. In addition, the system must be dried afterward. This method is more often used on systems that carry water or water vapor/steam during normal use, but it can be effective in cleaning gas lines as well.
Pigging, which is the practice of mechanically moving special devices through a section of pipe, is often used for cleaning purposes and is common for cleaning large pipelines in the petroleum industry. Pigging the gas supply lines at a power plant is slower and less effective than other methods, so it is typically used only in specific sections of the plant, such as the line up to the gas yard.
Moving on from Methane
In addition to the CSB and NFPA recommendations, in December 2011, the Electric Power Research Institute released detailed guidelines for fuel gas line cleaning using nitrogen or compressed air, upon which some of this article is based. In light of these developments, it is clear that industry momentum is moving away from the use of natural gas as a cleaning medium. Embracing the use of nitrogen and compressed air should lead to safer operations over the long term and help prevent future disasters like the Kleen Energy explosion.
—Thomas W. Overton, JD is POWER’s gas technology editor. Follow Tom on Twitter.