Lab-scale experiments have shown that seawater and calcium could effectively remove most of the carbon dioxide (CO2) from a natural gas power plant’s flue gas stream. A large fraction of the captured gas could then be converted into dissolved calcium bicarbonate—which, pumped into the sea, could be beneficial to the ocean’s marine life, says a researcher representing both the Lawrence Livermore National Laboratory’s (LLNL’s) Carbon Management Program and the University of California, Santa Cruz.
The results mean that above-ground CO2 hydration and mineral carbonate scrubbing may provide a “relatively simple” point-source CO2 capture and storage (CCS) system at coastal locations, reported Greg Rau in the February 2011 issue of Environmental Science & Technology. Rau, a senior scientist with the Institute of Marine Sciences at UC Santa Cruz who also works at LLNL, says, “Such low-tech CO2 mitigation could be especially relevant for retrofitting to existing power plants and for deployment in the developing world, the primary source of future CO2 emissions.”
Rau used a hollow, cylindrical Plexiglas reactor filled with between 2.2 kilograms (kg) and 2.5 kg of mineral carbonate aggregate of two test types: aragonitic coral fragments and calcitic limestone from a local quarry. The reactor was modified to allow seawater and gas streams to longitudinally flow though it. After full equilibration with air, Rau found that 85% of the captured carbon was retained in solution—that is, it did not degas or precipitate.
When added to the ocean, the alkaline solution resulting from the scrubbing process could potentially benefit marine ecosystems that are currently threatened by acidification, Rau further suggests (Figure 6). This would have the added benefit of using the “vast potential of the sea to safely sequester anthropogenic carbon,” he says. “The experiment in effect mimics and speeds up nature’s own process. Given enough time, carbonate mineral (limestone) weathering will naturally consume most anthropogenic CO2. Why not speed this up where it’s cost effective to do so?”
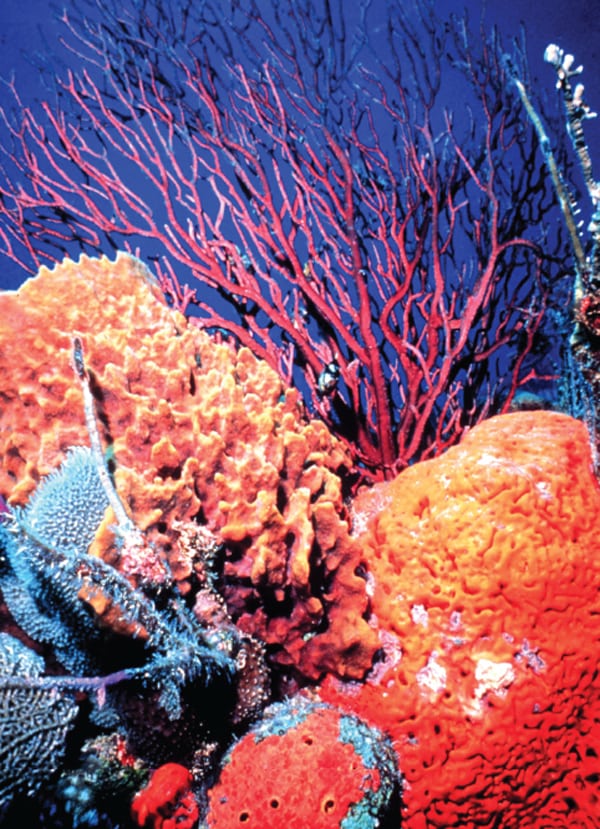 |
| 6. Alleviating acidification with flue gas carbon. A lab-scale experiment conducted by Greg Rau, a scientist from the Institute of Marine Sciences at the University of California, Santa Cruz and the Lawrence Livermore National Laboratory, suggests that seawater and calcium could effectively capture carbon dioxide from power plant flue gas. The resulting alkaline solution of dissolved calcium bicarbonate could then be pumped into the sea, alleviating ocean acidification. Source: National Oceanic and Atmospheric Administration |
Rau asserts that if the system converts and stores 80% of CO2 as calcium bicarbonate, it would cost less than $38/metric ton of CO2 mitigated. This compares with the more than $75/metric ton of CO2 mitigated that is estimated for CCS at conventional power plants. One reason for the cost advantage is that the limestone system avoids “CCS’s expensive capture and purification of molecular CO2,” he says.
The “relative technological simplicity” of such a system makes it ideal for rapid deployment in the developing world, Rau argues. He admits, however, that the method’s large demand for carbonate mineral and water would likely limit its application to coastal sites and that more research is needed to evaluate the system’s economics, potential scale, and permanence.
–Sonal Patel is POWER’s senior writer.