An economic evaluation plus real-world analysis of plants that converted from lime-based flue gas treatment to sodium bicarbonate treatment found that although lime systems are more familiar, sodium bicarbonate systems can be more economic for some waste-to-energy (WTE) plants when all costs and operating scenarios are considered.
Sodium bicarbonate is an adsorbent that has been used for a relatively short time in industrial flue gas treatment (FGT) processes. This additive is especially interesting for operators of smaller waste-to-energy (WTE) facilities.
In order to compare the use of sodium bicarbonate and lime hydrate, which is usually applied in (conditioned) dry flue gas cleaning systems, a dual approach was used. The experience and data from practical operation of plants that had, after retrofitting, experiences with both additives was one component. The other involved feeding data from the literature on the issue into an economic model that is able to consider a variety of different boundary conditions. With this dual approach, two scenarios were calculated and the influence of the different framework conditions on the results was evaluated using a sensitivity analysis.
Background and Motivation
The most common systems for removing acidic reacting components from flue gases of power plants—particularly sulfur oxides, hydrogen fluoride, and chloride—are alkaline scrubbers, often operated with lime components as neutralizing agent. Dry and semi-dry techniques on the basis of lime—predominantly Ca(OH)2—are also established, especially for smaller installations. But big power plants such as the 1,100-MW coal-fired Dry Fork power plant in Gillette, Wyoming, also are operated with dry lime-based methods.
A relatively new approach is the use of sodium bicarbonate (NaHCO3), also known as baking soda, as alkaline reagent for dry FGT. Although the price for sodium bicarbonate is significantly higher than for lime hydrate, this reactant has some advantages that make it interesting for use in flue gas cleaning. This is especially true for WTE plants incinerating municipal solid waste, waste wood, refuse-derived fuel, or sewage sludge, because of their normally limited size.
In Europe, especially in Germany, several operators of WTE plants decided in recent years to replace their existing flue gas cleaning system with a dry process based on the use of sodium bicarbonate. This stimulated a controversial debate about which process or additive is better, particularly more economical. In order to answer this question, a variety of comparative studies were designed. The comparison was usually carried out by doing exemplary model calculations based on typical operating parameters.
No practical experience from plant operation has been taken into account in any of the studies so far. For example, differences in operational management, unit availability, and personnel and maintenance efforts have not been considered. The study described here was intended to make a first contribution to addressing this deficit.
Chemical Reactions and Process Conditions
The use of sodium bicarbonate as an adsorbent for acid gases—instead of lime—does not require any conditioning of the flue gas, such as increasing the relative humidity in the exhaust gas. The deposition occurs as a gas-solid reaction, usually at temperatures of 180C to 200C and residence times of less than 2 seconds.
The sodium bicarbonate is first ground to increase its active surface area and injected directly after the mill into the hot exhaust stream. There the thermal decomposition, also called activation, takes place:
2 NaHCO3 → Na2CO3 + CO2 + H2O
This reaction can be observed, to a smaller extent, beginning with temperatures of around 60C. A highly porous structure, which provides a large surface area and thus ensures a high level of activity for further reactions of the produced Na2CO3 arises through the degassing of CO2 and water. Figure 1 shows scanning electron microscope recordings of surfaces of bicarbonate particles before activation, and Na2CO3, which occurred during activation.
 |
| 1. Before and after. Scanning electron microscope pictures of a NaHCO3 particle before (left) and a Na2CO3 particle after (right) thermal activation. Courtesy: Solvay |
Even if a certain proportion of the acidic components directly react with the bicarbonate, about 85% of the adsorbent is decomposed. Therefore the actual reaction partner is soda-ash:
Na2CO3 + 2 HCl → 2 NaCl + CO2 + H2O
Na2CO3 + 2 HF→ 2 NaF + CO2 + H2O
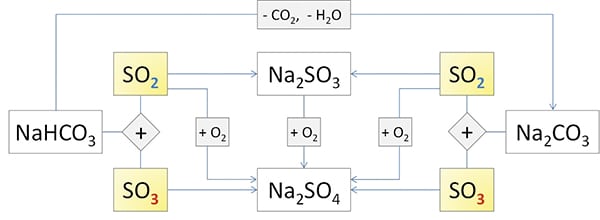
The reaction pathways for the reduction of sulfur oxides are more complex and are visualized in Figure 2.
The reaction of Na2CO3 with SO2 is highly temperature-dependent and can be described using the Arrhenius equation. Therefore, the reaction rate can be almost tripled by increasing the temperature from 160C to 220C. However, this behavior at temperatures above 400C is limited by sintering processes within the grain (the surface structure of the particles). This sintering affects both the sodium carbonate and its reaction products.
The reaction is limited with larger particle diameters, above 30µm, by pore diffusion.
Performance of Bicarbonate Compared to Lime
As noted above, most of the studies comparing lime and bicarbonate are model calculations based on assumptions. Operating costs—consisting of additives, residue management, energy (electricity, gas, steam, compressed air) and investment costs—are taken into account. Proceeds from the commercial provision of heat were also considered.
The results of such studies differ, according to selected approach and boundary conditions. Nevertheless, there are some common trends:
- Lime hydrate is often described as the more economical alternative.
- Considering energy efficiency, effort of retrofitting, operation and handling, and residue management, sodium bicarbonate shows advantages.
- According to the literature, the economic advantage of lime hydrate for plants equipped with selective noncatalytic reduction (SNCR) technology is greater.
- For waste incineration plants equipped with a DeNOx catalyst, the differences are said to be marginal, as the use of sodium bicarbonate allows an almost isothermal operation of the flue gas tract, and no energy costs arise from the reheating of flue gas before the catalyst.
Real-World Study Approach
Our investigation involved the examination of plants that had, after retrofitting, experiences with both sodium bicarbonate and lime hydrate systems. The operators were several times thoroughly interviewed about their opinions, and all relevant operational data (such as additive and energy consumption, emission values, downtimes, and maintenance effort) was, if available, obtained in detail and analyzed.
On the basis of this practically validated data, an economic analysis and comparison of the two processes and adsorbents was carried out, completed by a sensitivity analysis for all relevant parameters.
With WTE plants (at least in Germany), it is common to refer the consumptions of waste when describing capacity, because energy delivery is influenced by many (not only technical) factors. Waste incinerated in both plants is predominantly household waste, with significant amounts of commercial waste. The capacity of Plant SD is about 110,000 tons/year; Plant WS’s capacity is about 370,000 tons/year. Plant SD delivers about 50,000 MWh of electricity and 40,000 MWh of district heating each year to third parties.
Plant WS has an electric output of 35 MW. A figure for heat is not available because the produced steam is delivered to a nearby coal power plant that produces electricity with its turbines and uses a varying share of the steam for district heating, which is not separately accounted for.
Participating Plants
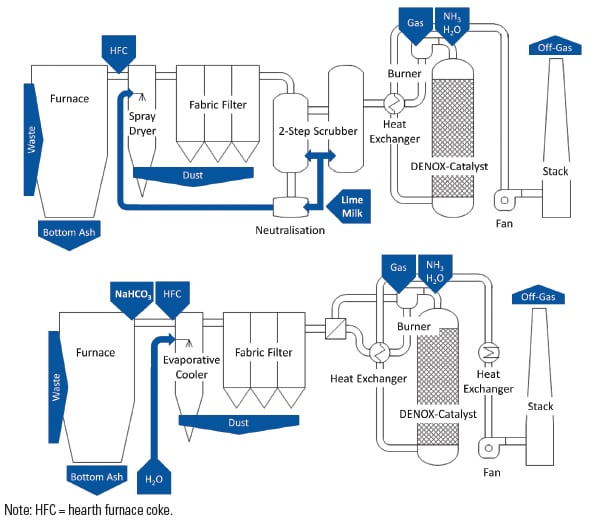
One of the plants participating in the study was equipped with a wet scrubbing system before reconstruction to the sodium bicarbonate process (Plant WS); the other one (Plant SD) had a semi-dry flue gas cleaning process (Figure 3).
  |
| 3. Participating plants. Plant WS is on the top; on the bottom is Plant SD. Courtesy: Plants WS and SD |
Figures 4 and 5 give an impression of the plants’ layout before and after reconstruction.
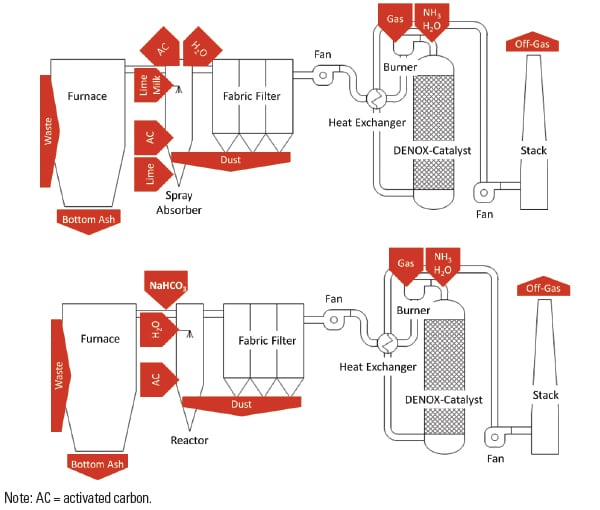 |
| 4. Process schemes for Plant SD. These diagrams show the plant before (top) and after reconstruction (bottom) to dry flue gas cleaning with sodium bicarbonate. Source: Dr. Peter Quicker et al. |
Plant SD was one of the first German waste incineration plants to use sodium bicarbonate for FGT. It was retrofitted in 2005 from a two-step conditioned semi-dry flue gas treatment with a spray absorber and additional dry lime hydrate injection to operation with sodium bicarbonate. The technical components largely continued to be used.
The three lines at Plant WS were retrofitted in 2010/2011 to operation with sodium bicarbonate. Before retrofitting, the plant was operated with a classic two-step washer system with an upstream spray dryer to evaporate the wastewater and a fabric filter to separate the evaporation products and other particles in the flue gas.
At both plants, operators were very satisfied with the retrofitting process. The estimated time schedule could be met in both cases. There were no significant problems with legal or technical issues, and initial operation did not cause any difficulties.
Plant Operation
According to the staff questioned, operation of the plants with sodium bicarbonate is considered to be positive in almost every aspect and an improvement compared to the system used before. The financial expectations could be met, in some cases even exceeded, mainly due to drastically reduced natural gas demand for reheating of the flue gas upstream of the catalyst.
Furthermore, significant savings could be achieved due to less maintenance and repair costs. An increase in availability and a reduction in personnel expenditure could be noted. Staff emphasized that the accident risk with handling sodium bicarbonate is minimal compared to highly caustic lime components. Due to these advantages, acceptance by personnel is said to be very high.
The weaker buffer effect of sodium bicarbonate is considered disadvantageous. This applies both in comparison with the washer operation as well as with conditioned dry flue gas treatment via lime hydrate. Nevertheless, emission peaks are handled without any problem.
Table 1 shows important operational parameters of both plants.
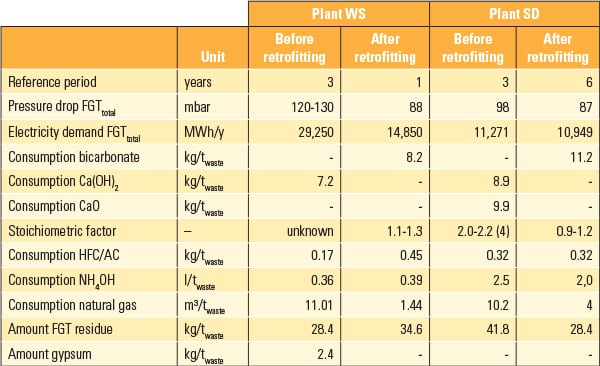 |
| Table 1. Important operational parameters for both plants. Source: Dr. Peter Quicker, et al. |
Additive Consumption
Due to the chemical valence of calcium (II), only half of the amount of calcium-based reagents is needed compared to adsorbents on a sodium basis (valence I) to bind the same amount of pollutant molecules in the flue gas, if complete sorbent utilization is assumed. As the sorption agent does not react completely and, in the case of lime hydrate, intermediate products (Ca(OH)Cl) are formed and side reactions (to CaCO3) take place, more of the reagent is needed in practical operation than in theory.
The stoichiometric factor of lime hydrate in dry applications is constantly above 1.6 and in the case of spray adsorption methods with lime milk is more than 2.3. Stoichiometric factors of 3 and even considerably higher may also occur. In Plant SD, stoichiometric factors between 2.0 and 2.2 were run at two-step (modified) conditioned dry sorption. At peaks, values up to 4 were reached.
Washers show stoichiometry between 1.0 and 1.1 as the additive is used for the pH value adjustment of the washing fluid and not directly as a reaction partner. For Plant WS, insufficient data were available to calculate the stoichiometric factors before retrofitting.
In the case of sodium bicarbonate, the stoichiometric factors are considerably lower than for lime hydrate. Published values are usually between 1.1 and 1.3. Only in exceptional cases are values higher than 1.4 given. The stoichiometric factors, determined as part of the study, for the use of bicarbonate of all investigated samples were: Plant SD—1.17 and Plant WS—1.26.
Considering the sodium contents already present in the flue gas, which could be quantified by the analysis of boiler ashes, considerably lower stoichiometric factors result, which, in the case of Plant SD, are even below 1.
Residues
Compared to the use of lime hydrate, the use of sodium bicarbonate leads to less residue, despite the fact that stoichiometrically a higher additive amount is needed. The reason is the separation of CO2 and water vapor during the activation reaction and the reaction with acidic gas components and the lower stoichiometric factors.
Data from Plant SD support this statement: Through retrofitting, the amount of residue was reduced from 42 kilograms/ton (kg/t) to 28 kg/t incinerated waste.
As a result of the low stoichiometric factors, the wet washing method results in the least residue. The amount of residue in Plant WS increased from about 28 kg/t waste up to 35 kg/t waste after reconstruction.
Energy and Efficiency
Energy efficiency in both plants was considerably improved through retrofitting. Crucial for this was a big reduction in natural gas consumption, which could be achieved because reheating of the flue gas up to catalyst operation temperature is no longer necessary. For Plant WS, referring to the average of the three years before retrofitting, a reduction in gas demand of 87% could be observed. In Plant SD, a reduction of about two-thirds, down to 3 m3/t waste in the last years could be achieved.
By retrofitting Plant WS, electricity consumption for flue gas cleaning and ventilation could be reduced from 29.2 GWh/year to 14.8 GWh/year. This is due to the reduction of pressure drop in the flue gas duct from 120–130 mbar to 90 mbar by removal of the scrubbers. The electricity demand for Plant SD did not change after retrofitting.
Emissions
The statutory emission limits were safely met by all considered methods, both before and after retrofitting. The effects on stack gas emission values resulting from the retrofitting can be described as moderate (Figure 6).
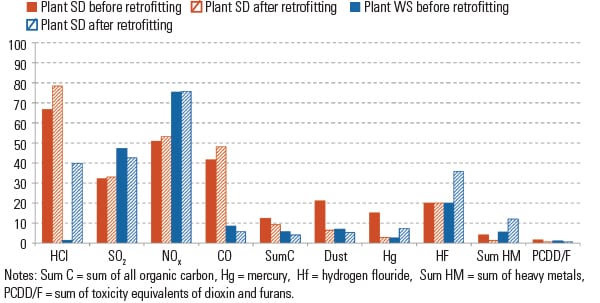 |
| 6. Emissions before and after retrofitting. This chart shows annual emission values as a percentage of the legal limit. Source: Dr. Peter Quicker et al. |
As to be expected, the efficiency of the HCl separation in Plant WS after retrofitting from the washer system to dry flue gas treatment was reduced by more than the power of 10. The increase in clean gas emissions for the parameter hydrogen fluoride (HF), which is a result of less affinity of sodium bicarbonate towards hydrogen fluorite, is also known.
Economic Comparison
An economic comparison of the dry sodium bicarbonate and the conditioned dry lime hydrate processes was carried out. Scrubber systems were not included, because they are currently not relevant for the German market.
In order to grasp the importance of different framework conditions regarding the economic efficiency of the methods, a calculation algorithm was developed that is able to include (besides the usual parameters of economic calculations—investment, additive, disposal, and energy costs) additional parameters identified as relevant after discussions with the operators. There is, for example, the option to specify higher availabilities due to longer maintenance intervals or less-frequent downtimes, to integrate additional revenues through seepage water treatment, or to investigate different settings for optimized heat management.
Two operation points have been considered as basic scenarios, where the additional options previously discussed have initially not been taken into account. One is a very conservative approach. To achieve better comparability, this approach is geared to the assumptions and framework conditions of a study on the economic comparison of dry sorption carried out for the German Lime Association. The second is an approach described as “practical,” for which a higher stoichiometric factor for the use of lime and an alternative residue composition (ratio Ca(OH)Cl: CaCl2) has been considered.
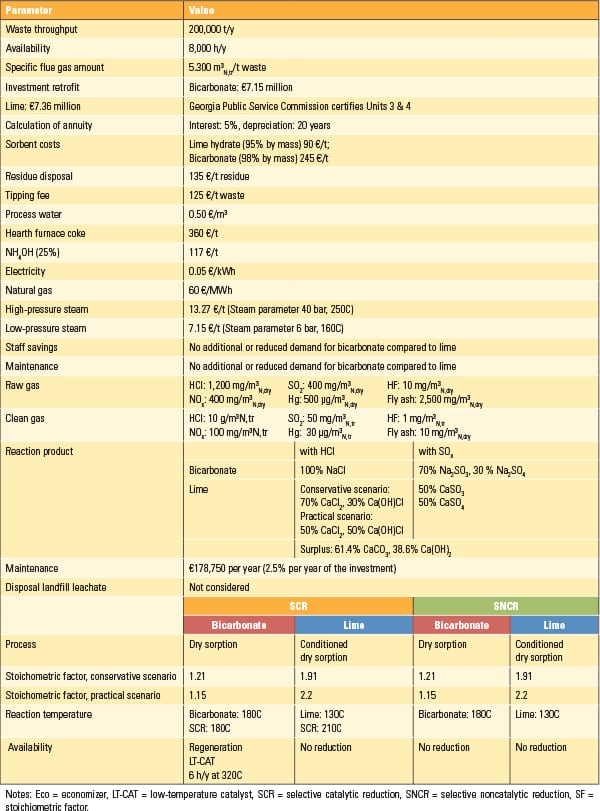
Assumptions for economic calculations are summarized in Table 2, available in the online version of this article.
 |
|
Table 2. Assumptions for economic comparison of lime- and bicarbonate-based flue gas treatment. Source: Dr. Peter Quicker et al. |
Results of the calculation, differentiated by the two scenarios (conservative and practical) and by the kind of the NOx reduction process, are listed in Table 3.
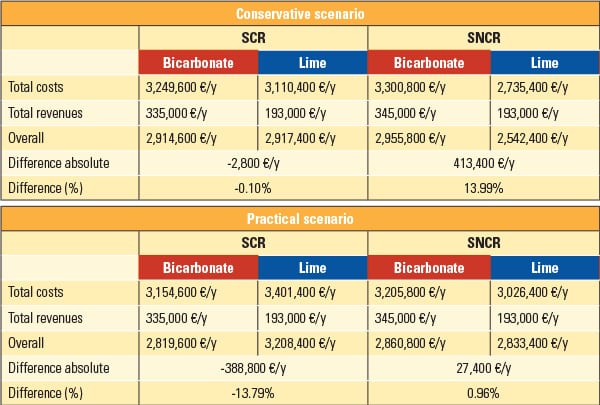 |
| Table 3. Results of the economic comparison of lime and bicarbonate. Source: Dr. Peter Quicker et al. |
It has been shown that the conservative basic scenario with selective catalytic reduction (SCR) technology leads to the same overall result for both additives. The higher price of bicarbonate is compensated for by the energy demand required for reheating the flue gases, which is necessary when using lime hydrate. If a natural gas duct burner would be used instead of a vapor gas preheater for heating the flue gas, an annual cost disadvantage of approximately €750,000 would be noted when using lime hydrate. If the scenario described as practical is considered, a significant cost advantage of 13.8% for the operation with bicarbonate is achieved.
For SNCR technology, flue gas reheating before the catalyst does not occur, and the main disadvantage regarding energy efficiency of the method using lime hydrate is not relevant. In this case, the conditioned dry sorption with lime hydrate is therefore superior to the method using bicarbonate. Calculations for the conservative scenario lead to a cost advantage of about €413,400/year, or 14%. Considering the practical scenario, the costs for both methods, using bicarbonate or lime hydrate, are about the same.
Both the “conservative” and “practical” scenarios were used as reference points for the sensitivity analysis of different framework conditions. Using this analysis, parameters relevant for the economic efficiency can be identified as well as those that are of less importance.
It has been shown that investment costs have the lowest effect on the difference between the relative costs of both treatment options. The following parameters basically have a positive influence on the economic efficiency of the bicarbonate method, compared to the lime hydrate process:
- Low pollutant concentration in the exhaust gas
- Dropping additive costs
- Increasing energy prices
- Increasing disposal costs
Of course, an increase in availability also has positive effects. Elimination of the gas-fired reheating of exhaust gas upstream of the catalyst has the strongest influence on economic efficiency. This is fully supported by the experiences of the participating operators.
Positive Reception for Sodium Bicarbonate
The operators and personnel of both plants participating in the study consistently assess the process of retrofitting and subsequent operation with sodium bicarbonate as very positive. Acceptance is high. Significant savings through the elimination of maintenance and repair costs as well as an increase in availability and a reduction in personnel hours could be noted. Quantification of these effects is difficult because of the quality of available data, so generalizations cannot be made.
In both cases, financial expectations of the retrofits were fulfilled, and partly even exceeded, mainly because of drastically reduced gas demand resulting from the elimination of exhaust gas reheating before the catalyst.
The conducted conservative economic calculation results in comparable total costs for FGT with lime hydrate and sodium bicarbonate, when the regarded plant is equipped with a DeNOx catalyst. If SNCR technology is in use, the option with lime hydrate has economic advantages for the conservative scenario.
Through the sensitivity analysis, parameters were identified that have a significant influence on economic efficiency. For instance, the increase in unit availability can contribute significantly to improved economic efficiency.
Altogether, the use of sodium bicarbonate as an adsorbent for FGT is recommended especially at plants with the following conditions:
- SCR technology.
- Rising energy costs.
- Possibilities for commercial provision of heat.
- High disposal costs for residue from flue gas cleaning (possible recycling interesting).
- Low concentrations of acidic hazardous gases (especially HCl).
The choice of suitable FGT methods for new or retrofit plants also requires site-specific considerations. ■
— Dr. Peter Quicker is a professor in the Unit of Technology of Fuels RWTH Aachen University, Aachen, Germany (info@teer.rwth-aachen.de). Engineers Martin Rotheut, Yves Noel, Marc Schulten, and Uwe Athmann contributed to the research and writing of this article.