In Part I of this two-part report we examined the various chemical forces at work in condenser tube leaks, the steam plant components placed at risk, and the suite of instrumentation most capable of providing early warning of a leak. Assuming you were able to repair the leak and quickly resume operation, the next step is to identify the damage mechanisms that caused the problem so you can minimize future leaks.
Condenser tube leaks are caused by corrosion or damage mechanisms that affect the entire condenser. Those mechanisms will continue to cause additional leaks until the root cause(s) of the failures are identified and changes are made to materials, water treatment, or plant operations to eliminate them. This article describes some of the more common condenser tube failure mechanisms and provides a brief discussion of how they might be prevented.
The first step is to obtain a sample of the tube that failed for metallurgical examination to determine the failure mode. Nondestructive evaluation (NDE) techniques, such as eddy current testing, may be needed to provide an indication of the scope of the damage, but they cannot pinpoint a failure mechanism. Together, metallurgical testing plus some type of NDE can lead you to the root cause of the failure.
Condenser Tube Materials
Common condenser tube materials include various grades of copper brasses such as admiralty brass, 90:10 copper-nickel (Cu-Ni), and 70:30 Cu-Ni. In copper alloy condensers, it is not uncommon to find higher-grade materials, such as stainless steel tubes, used in the air removal section.
Series 300 stainless steel alloys, such as 304 and 316L, are now fairly common in new construction in both freshwater and seawater applications. Higher-grade duplex stainless steels, such as AL6XN and 6 Mo stainless steel alloys, may also be used. Superferritic stainless steel, such as Sea-Cure, has also been used for cooling water where high levels of chloride are present. These improved materials are required when using brackish and other alternative water sources.
The duplex and superferritic stainless steels are far more resistant to stress corrosion cracking than the Series 300 alloys. The table lists stainless steel materials with their Pitting Resistance Equivalent Number (PREN) and Critical Crevice Corrosion Temperature (CCCT). The maximum chloride value is the value below which chloride crevice corrosion will not occur.
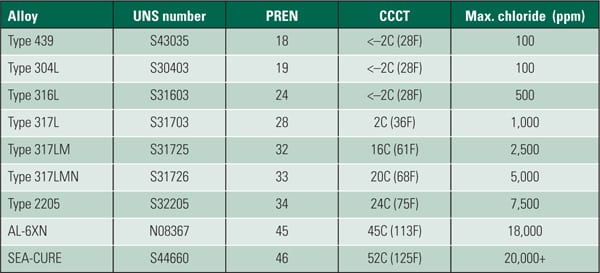 |
| Corrosion and pitting resistance of various stainless steel alloys. Source: J.C. Tverberg, “Performance of Superferritic Stainless Steels in High Chloride Waters,” presented at Condenser Technology: Seminar and Conference, EPRI, Palo Alto, Calif., 2002 |
The standardized testing conditions under which these values were developed are much worse than conditions normally experienced using traditional sources of cooling water. The best approach is to use this information as a comparison of the relative corrosion resistance of one alloy against another, not to set limits for cooling water chemistry. Not shown in the table are titanium alloys that are also used in seawater applications or when the cooling water supply is high in chlorides.
Each of these materials has its advantages and disadvantages. As noted above, certain stainless steel alloys are more resistant to chloride pitting and corrosion than other alloys. Copper alloys, such as 90:10 and 70:30, though often discounted due to their lower resistance to erosion, have a natural resistance to microbiologically influenced corrosion (MIC) caused by the toxicity of copper ions to many living species. However, this same toxicity may create a problem if the plant’s cooling water discharge is returned to a public waterway. In such cases, a plant may be required to maintain the copper concentration of the discharged cooling water below very low levels.
Often, when retubing an existing condenser, the material selection may be limited due to the lower heat transfer coefficients of stainless steel and titanium alloys compared with copper alloys. Titanium alloys, though typically considered immune to corrosion from the cooling water side, have failed due to impingement from saturated steam dump lines in the condenser. They can also suffer from failures if steel tools scratch and penetrate the protective titanium oxide layer.
Common Condenser Tube Failure Mechanisms
In our experience, the most common causes of condenser tube failure are related to erosion, corrosion, or some form of mechanical damage. The following discussion is meant to guide your initial examination of the failed tubes and to help you close in on the root cause.
Erosion. Cooling water erosion of condenser tubes occurs in areas of high turbulence and velocity such as in the first few inches of the inlet to the condenser tube, or in areas where there is an anomaly in the tube, such as a deposit or foreign object that the water must flow around. Figure 1 shows the latter case.
 |
| 1. Cratered brass. Admiralty brass tubing that carries the cooling water in a condenser can erode because of excessive water velocities or because the water is contaminated. Courtesy: M&M Engineering |
Erosion is worse during periods of high flow rates or when sand, fly ash, or other abrasive materials are entrained in the water. High localized flow rates can occur when the tube sheet is partially blocked by debris. Copper-nickel alloys have twice the resistance to erosion of admiralty brass. But there is at least an order of magnitude difference between copper-nickel alloys and stainless steel or titanium alloys. Erosion can also affect the tube sheet and create leaks in the crevice between the tube and tube sheet.
Erosion can also occur from the steam side. Emergency dump lines or drain headers can impinge on the condenser tubing below in a very localized area. In one case, the proximity of a reheater dump header caused a high-velocity water and steam mixture to impinge on the top row of a condenser with titanium tubes (Figure 2). The plant is seawater-cooled, and the resulting tube leak caused massive contamination of the turbine requiring an extended outage to clean and inspect the turbine and associated valves and piping. Erosion of support plates may also increase the propensity for vibration damage.
 |
| 2. Leaky titanium tubes. Wet steam caused the erosion of these titanium condenser tubes. Courtesy: M&M Engineering |
Erosion on the waterside can be minimized by keeping the tube sheet and tubes clean and free of debris and operating the cooling water flow rates within the design range. Regular inspection can detect waterside erosion. Tube inserts, both metal and plastic, have been used to protect the first few inches of the condenser tube from further erosion and leaks.
Care should be taken when using very high pressure cleaning equipment on condenser tubes. Excessive pressure or inexperienced operators can damage tubes and create more problems than they solve.
Regular and careful visual inspection of the steam side of the condenser should locate areas where steam erosion is occurring. Steam/water shields can then be installed to deflect impingement, or the affected tubes can be plugged or replaced with a more erosion-resistant alloy.
Waterside Corrosion. One of the most common cooling water–side corrosion mechanisms in condenser tubing is microbiologically influenced corrosion (Figure 3). It is far too common in stainless steel tubing and less common, but still possible, in copper alloy tubing. A very few cases of corrosion in titanium tubes have even been ascribed to MIC.
 |
| 3. MIC damage. This stainless steel condenser tubing was attacked by microbiologically influenced corrosion (MIC). Courtesy: M&M Engineering |
Stainless steel condenser tubing is protected from corrosion by a thin layer of metal oxide. When a biofilm forms over the tube, the chemistry between the biofilm and the condenser tube often becomes corrosive to the oxide layer and underlying metal. The more well-established, thick, and microbiologically diverse the biofilm is, the more likely it is that MIC can form underneath it.
In nearly any water-containing system—and certainly in cooling towers and once-through cooling applications—biofilms cannot be eliminated; they can only be controlled. (See “Biofouling Control Options for Cooling Systems,” September 2007 in our online archives at https://www.powermag.com.)
The most common control strategy for biofilm in cooling water is the use of bleach to produce hypochlorous acid, an effective biocide. In some cases the bleach is combined with sodium bromide to produce hypobromous acid, which is a better biocide in alkaline pH cooling water. In cooling towers, the effectiveness of bleach or bleach/bromide can be enhanced with biodispersants. Bleach or bleach/bromide, applied properly and consistently, will typically provide adequate control of biofilms and minimize MIC.
Mechanical cleaning, whether with brushes, scrapers, foam balls, or high-pressure water lancing, is the only cleaning technique that can effectively remove all biofilm in a condenser tube. Units that regularly use one or more of these mechanical cleaning methods are far less likely to see condenser tube failures due to MIC or other issues with biofilm, regardless of the biocide program that is in use.
Titanium tubes have a very tenacious and chemically inert passivation layer that protects them from nearly all corrosion mechanisms. It is very difficult to accumulate enough of the right kind of bacteria in a conventional power plant cooling system to create an environment that would be corrosive to titanium oxide. However, titanium tubes are still subject to biofilm accumulations that can slow the flow through a condenser and cause problems in the cooling tower when biofilm accumulates on the fill.
Steamside Corrosion. Corrosion is also often found on the steamside of a condenser tube. Admiralty brass tubing is susceptible to both ammonia grooving and ammonia-induced stress corrosion cracking.
Ammonia grooving occurs when ammonia added for pH control of the feedwater and carbon dioxide and oxygen from air in-leakage condense with the steam, run down the inside of the tube sheet or other support, and cause corrosion in the crevice between the tube and tube sheet (Figure 4). Although the focus is often on reducing ammonia (and therefore the pH of the feedwater), the real culprit is the air in-leakage, which brings in the carbon dioxide and oxygen. Both carbon dioxide and oxygen are essential to the corrosion process. This is why the air removal sections of admiralty condensers often contain stainless steel tubes.
 |
| 4. Groovy tubes. Too much ammonia in the cooling water caused grooving on both sides of a condenser tube support. Courtesy: Andy Howell, Xcel Energy |
Carbon dioxide also lowers the pH of the condensate, requiring additional ammonia feed to maintain the desired pH in the feedwater, producing higher ammonia levels in the condensate. High levels of ammonia can also cause stress corrosion cracking in admiralty brass (Figure 5).
 |
| 5. Admiralty attacked. Excess ammonia in the cooling water can also cause stress corrosion cracking of admiralty brass tubing. Courtesy: Andy Howell, Xcel Energy |
Susceptibility to ammonia grooving and ammonia-induced stress corrosion cracking drops dramatically as the amount of nickel in the alloy increases. Even a 90:10 Cu-Ni alloy provides far more protection against these forms of corrosion.
Series 300 stainless steel alloys are susceptible to chloride-induced stress corrosion cracking, though a serious contamination event and a concentration mechanism (crevice or wet/dry conditions) would normally be required to create chloride-induced stress corrosion cracking on the steam side of a condenser tube.
Stainless steel alloys are also susceptible to enoblement corrosion. This form of corrosion results from the combined action of manganese in the water source, bacteria, and the action of bleach or chlorine. Manganese can be found in seawater, lakes, and rivers. In freshwater sources, the levels of manganese can spike seasonally with lake turnover or spring runoff. Bacteria in biofilms on the surface of the condenser tubes can collect and precipitate the manganese and create a black magnesium oxide coating. This coating can create an uneven deposit layer that is more noble than the stainless steel, making it the anode and thus causing pitting corrosion.
Other Failure Modes
Mechanical vibration can result in catastrophic condenser tube failures or leaks that are very difficult to find because they open and close depending on steam conditions. The number of tube failures due to vibration may increase suddenly after a turbine upgrade that uses more steam or creates a different set of harmonic vibrations in the condenser.
When considering a turbine upgrade, it is good to examine the condenser design to ensure that the changed steam flow rate doesn’t create areas of mid-span collisions, fretting, or fatigue in the condenser tubes. Regular visual inspection of tube supports, NDE, and modeling of the condenser with computational fluid dynamics analysis are all tools that can be used to determine the potential for failures due to vibration.
Plugging of the tube sheet with debris and macro-biological fouling (such as from fish or clams) can be problematic for units using cooling water from lakes, rivers, or seawater. A small tear in a screen can result in a large accumulation of debris on the tube sheet that can plug a significant number of the condenser tubes, forcing the remaining water through the remaining tubes at higher velocities. The plugged or partially plugged tubes can develop calcium carbonate deposits and silt accumulations due to slower flows. These deposits can then become havens for microbiologically influenced corrosion.
Freeze protection is also important for any condenser where temperatures drop below freezing or any using cooling water that can freeze. Some water boxes may not drain completely, particularly if the drain is partially blocked or rusted shut. Figure 6 shows a titanium tube that failed due to freezing temperatures.
 |
| 6. Frozen solid. This titanium condenser tube failed when water left in the tube froze. Courtesy: Andy Howell, Xcel Energy |
Special thanks to Ray Post of ChemTreat and Andy Howell of Xcel Energy for their contributions to this article.
— David G. Daniels (david_daniels@mmengineering.com) is a principal of M&M Engineering and a POWER contributing editor.


















