Corrosion is a constant battle for infrastructure of all kinds—the natural response of materials like metals to their environment, causing damage to everything from turbines to oil pipelines and vehicles. The costs are vast. A National Association of Corrosion Engineers (NACE) IMPACT study published in 2013 claims that corrosion causes damage valued at $2.5 trillion each year, or between 3% and 4% of annual gross domestic product (GDP) of developed nations; the result of corrosion causing enforced shutdowns, accidents, and “near misses.” These cost figures also haven’t taken into account the consequences of corrosion for the environment or impact on human health and safety.
In power generation, the challenges have been greatly intensified by demands for reductions in greenhouse gas emissions and the introduction of new fuels, such as hydrogen and low carbon biomass feedstocks. These have meant a substantial heightening of the intensity of environments, higher steam pressures and temperatures, that are ramping up the damage caused by corrosion through heat exchanger degradation and the formation of more and different corrosive deposits, some of which have yet to be studied in any great depth.
For the energy generation sector, the situation is demonstrated starkly by the damage being done to turbines involved, with traditional turbine blades costing £30,000 ($38,250) to replace, and single-crystal turbine blades above £100,000 ($127,500). The conventional approach to maintenance involves stoppages and the removal of a section of a turbine blade to assess levels of corrosion and any replacements needed (Figure 1).

State of Coatings
While corrosion mitigation is on the agenda for industry, much more could be done with proactive adoption of new technologies, as well as looking at ways to collaborate in order to share corrosion mitigation strategies and work to unify standards and practices—for the sake of the environment, safety, and good business. There is a real need to assess the potential for the use of different materials and coatings.
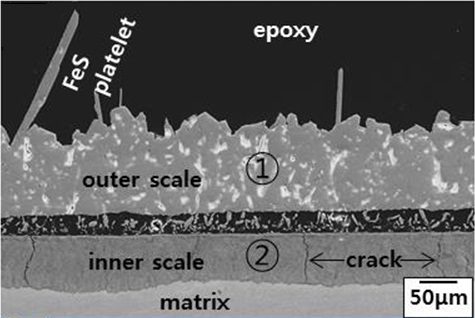
Currently, the response to the new environments caused by low-carbon fuels has been the use of expensive nickel-based materials or coatings. Our studies have looked at the behaviour of three different coatings subjected to the new biomass environment: NiCrFeSi (deposited via High Velocity Oxygen Fuel [HVOF], a thermal spray coating process used to improve a component’s surface), FeCrAl (through laser cladding—where a laser beam is used to deposit a coating of a stream of metallic powder or wire into a melt pool), and NiCrFeSi (also via a laser clad).
The three coatings were deposited onto tube segments of a stainless-steel alloy (347HFG)—a steel that could be used in superheaters/reheaters—and exposed to a simulated biomass combustion environment, involving a temperature of 700C for 1,000 hours. The resulting data showed how the three different coatings studied suffered enhanced corrosion attack, resulting in internal corrosion and “diffusion cell” type of behaviour. None of the three coatings were able to stop the diffusion of corrosive elements through the coating thickness, which also affected the substrate alloy. Among the three coatings, the one composed of NiCrFeSi deposited through laser clad seemed to be the only one remaining on the surface of the alloy, although it did suffer the same type of attack. The main reason for the slightly better performance of the NiCrFeSi deposited through laser clad could be attributed to the higher thickness of the coating.
Constant Evolution
At Cranfield University, we are working on simulation models that would allow businesses to accurately predict when the materials involved need to be repaired or replaced, minimising maintenance checks and materials’ waste, avoiding the need to carry out manual checks and cause damage. The emergence of new computing technologies, such as artificial intelligence (AI) and neural networks, are opening up new branches of research and development in materials science, which could lead to a better understanding of the properties of materials already in use, and in the design of new materials.
There is no ideal material that would work in any environment. Each application needs its own materials, and these need to evolve and adapt as demands and circumstances change in their particular environment. This is why corrosion mitigation and cost savings are dependent on constant experimentation and exploration. The need for collaboration and synergies between industries and universities is essential.
It will also be important to learn and borrow from other sectors. For example, in the marine industry there are new trends emerging around the design of smart coatings. In recent years, researchers have been working on the development of coatings with the ability to repair themselves once damaged (by loading the material with a healing agent in microcapsules or similar form that is released when the material is damaged by corrosion)—the kind of technology that would be hugely valuable if translated for use into the new world of high-temperature regimes.
—Stefano Mori, PhD is a lecturer in Energy and Materials at the Centre for Energy Engineering at Cranfield University (www.cranfield.ac.uk).