The University of Iowa (UI) Biomass Fuel Project has produced significant reductions in greenhouse gas emissions, regulated boiler stack emissions, and purchased energy costs. The project utilizes a renewable, biomass fuel source and provides an opportunity for UI to partner with a local industry.
UI was approached by Quaker Oats Co., Cedar Rapids Facility in late 2001 about using oat hulls produced in the food-making process as a source of fuel for UI’s circulating fluidized bed (CFB) boiler. Changing business conditions had caused Quaker to seek alternative outlets for the oat hulls. Because UI has a CFB and was located only 20 miles away, there appeared to be an opportunity to use the hulls as a source of biomass fuel.

1. University of Iowa’s power plant co-fires coal and oat hulls. Courtesy: University of Iowa
Over the next two years, a team of engineers and staff from UI, Quaker, and Foster Wheeler conducted test burns (see sidebar) and designed and installed a fuel feed system for the oat hulls. In addition, the CFB boiler was permitted to co-fire the oat hulls and coal. Full commercial operation of the project started in 2004.
The UI Power Plant’s partnership with Quaker Oats Co. helps the food manufacturer’s production operation by providing a reliable, stable market for oat hulls. UI benefits because the oat hulls are a renewable energy source and because burning oats hulls enables the university to reduce greenhouse gas emissions and regulated air pollutants from its power plant.
UI operates a combined heat and power facility with two solid fuel boilers, one CFB and one stoker, and two natural gas boilers. The plant has a total capacity of 580,000 lb/hr of 500 psig 750F steam. It supplies steam to 119 campus buildings and to the University of Iowa Hospital and Clinics. Three controlled extraction condensing steam turbines, with a capacity of 24.7 MW, economically and efficiently cogenerate about one-third of the total facility electric power needs.
CFB Boiler Operation
CFBs are noted for their ability to burn a wide variety of fuels. At UI the CFB was originally designed to burn 100% coal. In the CFB process, air is injected into the furnace with primary air (PA) and secondary air (SA) fans. Air flows from the fans though an air heater, where heat in the exhaust gas is recycled into the combustion process. The PA then flows into the bottom of the combustion chamber and up through a grid floor.
A grid floor fluidizes material in the furnace. Substiochiometric (oxygen lean) combustion occurs in the bed material just above the grid floor. As the bed material rises, secondary air is injected and the combustion process continues, and powered limestone is injected with SA. Calcium in the limestone chemically reacts with SO 2 to form calcium sulfate, or gypsum (CaSO 4), a solid material removed with the ash.
The bed material resembles beach sand with chocolate chips — that is, chunks of unburned coal. While the unit is operating, about 25 to 40 tons of material is in constant circulation in the boiler furnace. The sandlike bed material contains limestone, fuel, and ash. Most of the circulating bed material is limestone that has reacted with the sulfur in the coal.
As the furnace material rises in the furnace it becomes less dense. When it reaches the top of the furnace, it is passed through cyclones, where combustion gases and light ash are separated from the heavier material. All of the heavy material is returned to the bottom of the furnace above the grid floor. Coal is added before it is reinjected into the furnace. The light ash and boiler gases continue through the boiler heat recovery equipment, baghouse filter, induced draft (ID) fan, and boiler stack.
Limestone Consumption
Limestone consumption rate is a function of the fuel’s sulfur content, reactivity of the limestone, and furnace temperatures. Sufficient limestone is injected to ensure that a minimum of 90% of potential SO 2 emissions are removed. One measure of the effectiveness of a CFB furnace is the calcium to sulfur ratio (Ca:S). This is calculated by dividing the number of moles of calcium required to remove 1 mole of sulfur, through the reaction of converting SO 2 to CaSO 4. Another characteristic of CFB furnaces is that NO x emissions will typically increase with increasing limestone consumption.
Oat Hulls’ Attributes
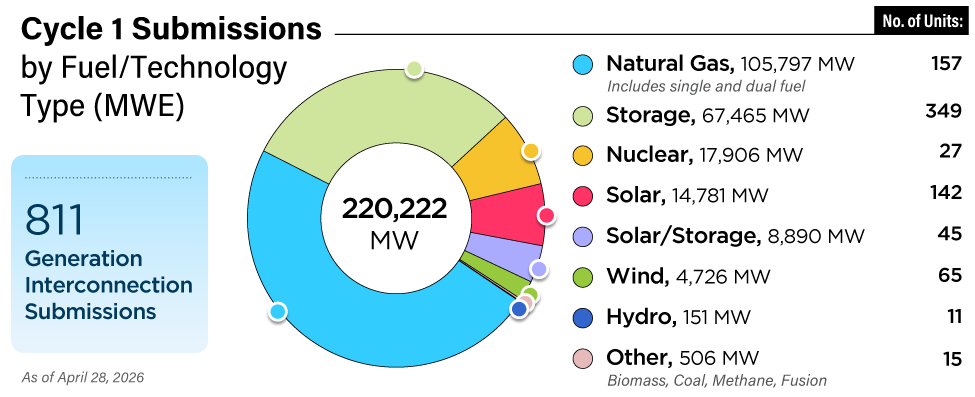
An oat hull is the outer shell of an oat grain that remains after the soft, protein-containing core has been removed by milling the grain. The oat hull has little protein but does have a heat content of about 7,000 Btu/lb. Coal used in the plant’s CFB boiler has a heat content of about 11,000 Btu/lb. Other significant differences between coal and oat hulls include the percentage of carbon, oxygen, sulfur, and material density. Figure 2 compares the ultimate analysis of oat hulls and CFB coal.
2. As-received ultimate fuel analysis of coal and oat hulls. Source: University of Iowa
The lower carbon content in oat hulls accounts for a lower heat content (Btu/lb) when compared with coal (44% versus 65%). In addition, oat hulls have a significantly higher percentage of oxygen compared with coal (35% versus 7.4%). Because of the oat hulls’ higher oxygen content, the plant’s boiler combustion control system had to be modified to reduce the amount of combustion air supplied to the furnace as the co-firing rate of oat hulls increased. Another benefit of using oat hulls is they have almost zero sulfur — an improvement over coal.
Oat hulls are about the size of sunflower seeds and lightweight — 7 lb/ft 3, whereas coal is 55 lb/ft 3. The cost of transporting this light material is a major factor in making a project like this economically viable. Fortunately, the Quaker Oats facility is only 20 miles away from the UI Power Plant.
Operating with Oat Hulls
Although the original plan was to co-fire oat hulls and coal, initial material-handling tests demonstrated that the existing coal system was not suitable for blending the two fuels. Additionally, design of the existing coal silos caused the oat hulls to bridge. As a result, they were unable to flow out of the silo. A new silo and separate lean phase pneumatic fuel injection system was designed and installed to convey oat hulls from the silo into the furnace.
The new oat hull silo has a mass flow design with a much greater angle of repose in the bottom. Oat hulls, which are lean phase, are transported and injected into the furnace through secondary air ports. When injected into the furnace, the oat hulls burn quite rapidly and the flame produced closely resembles a gas or oil flame.
Furnace erosion and accelerated wear in the CFB furnace was a concern due to the higher level of silica in oat hull ash. Silica in the oat hulls is over 70%. Since 1996 when the boiler was rebuilt, certain areas of the furnace water walls, low in the furnace and above the refractory line, always had accelerated wear. However, periodic furnace inspections and welding repairs were adequate to maintain satisfactory tube thickness.
Unfortunately, after the oat hull project started, higher water wall wear rates were observed during periodic furnace inspections. However, at this time it has not been determined if the accelerated wear is caused by increased silica levels circulating in the furnace from the oat hull ash or from operating the furnace at higher loads for longer periods of time.
In order to ensure maximum boiler availability, a solution to the accelerated wear was sought that would reduce furnace tube wear rates to acceptable levels. This was accomplished by replacing the lower 20 feet of the furnace water walls with shop-fabricated, metal flame-sprayed panels in the fall of 2005.
Prior to the oat hull project, the CFB typically operated below its maximum continuous rating (MCR). When co-firing coal and oat hulls the CFB now produces the lowest-cost steam and is operated at MCR almost continuously.
Emissions
In 2003 the existing CFB operating permit stated that only coal could be burned; any other fuel required permission from the issuing agency, the U.S. EPA. In order to process a permit revision allowing oat hulls to be burned in the CFB, the regulatory agencies responsible for UI — the EPA and Iowa Department of Natural Resources — needed combustion emissions data. Because no data were available that demonstrated how CFB emissions change as coal and oat hulls are burned in the same furnace, UI conducted extensive stack testing to characterize these emissions.
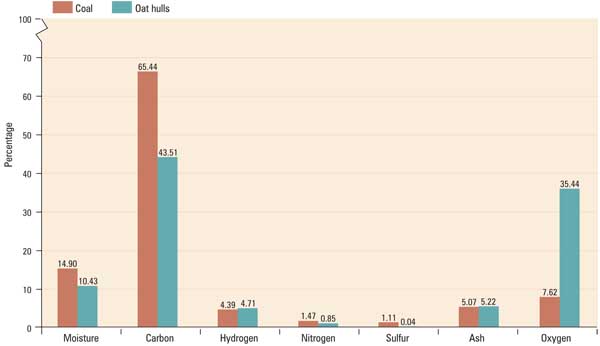
Three stack tests were performed, all with the boiler at maximum load. In the first test the unit was fired with 100% coal; the second test was performed with 50% heat input from oat hulls and 50% heat input from coal. The final run was conducted at an oat hull/coal heat input ratio of 80:20. SO 2 emissions were expected to decrease as the heat input from oat hulls increased, because oat hulls have virtually no sulfur content. Testing confirmed this hypothesis.
As expected, NO x emissions decreased when co-firing oat hulls with coal. Because less mass of sulfur must be removed to meet the 90% SO 2 reduction requirement, less limestone is required. The lower limestone consumption rate also reduced emissions of NO x. A stack test conducted at a 50/50 heat input from oat hulls and coal showed a decrease in NO x emissions.
However, as oat hull heat input increased from 50% to 80%, NO x emissions did not continue to decrease but remained constant. This is likely due to the Ca:S ratio increasing, as the amount of sulfur entering the furnace decreases with higher oat hull co-firing rates. Recent UI CFB process data analysis has confirmed that the Ca:S ratio increases as the amount of sulfur entering the furnace decreases when co-firing oat hulls.
Stack testing has demonstrated that particulate emissions decrease with increasing oat hull co-firing rates. This result was not anticipated. Because oat hull combustion produces flyash but no bottom ash, there was concern that particulate emissions would increase with rising oat hull firing rates. The additional flyash might also degrade the baghouse’s performance.
However, the opposite performance was observed as oat hull co-firing resulted in an initial decrease in particulate emissions at 50% heat input and an additional decrease at 80% heat input from oat hulls. One explanation for decreasing particulate emission with increasing heat input from oat hulls may be that the added dust loading on the baghouse results in improved filtering performance of the fabric filters.
As oat hull firing increased, a decrease in carbon monoxide emission rates was observed. The level of carbon monoxide emissions is indicative of complete combustion of the fuel. A possible explanation for the trend of decreasing carbon monoxide emissions with increasing oat hull firing rates is that the oat hulls are contributing to more complete combustion in the boiler. The table summarizes results of emissions stack testing.
Emissions stack testing results. Source: University of Iowa
Total hydrocarbon emissions are lower with biomass co-firing when compared with firing 100% coal. However, the reason for this, or the increase in hydrocarbon emissions at higher biomass firing rates, is not apparent at this time. More study is needed to understand this phenomenon.
Committed to Reducing Greenhouse Gases
In addition to reducing the regulated pollutants, displacing coal by burning a renewable, biomass fuel (oat hulls) reduces CO 2 emissions that would be produced had the displaced coal been burned. Each ton of biomass burned displaces more than a half ton of coal. Burning 1 ton of coal causes 2.5 tons of new CO 2 to go into the atmosphere. CO 2 is a greenhouse gas (GHG) that is believed to make a significant contribution to global warming. Reducing GHG emissions is a key part of UI’s environmental stewardship.
Burning oat hulls does produce CO 2 emissions through carbon combustion. But this carbon became part of the fuel through photosynthesis. Carbon in coal comes from fossil fuel located underground. Though the source of carbon in fossil fuel was originally plant material, it was formed millions of years ago.
When carbon in coal or any other underground fossil fuel source is burned, CO 2 released to the atmosphere increases the current global inventory of this GHG, whereas CO 2 produced by burning biomass does not increase the current CO 2 global inventory, because the carbon was already in the atmosphere before the oat (or other) plant grew.
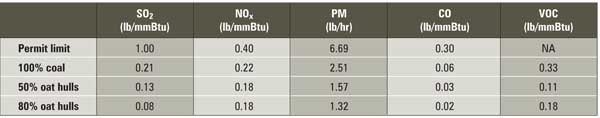
UI joined the Chicago Climate Exchange (CCX, www.chicagoclimateexchange.com) in 2004. Through its membership in CCX, the university made a commitment to reduce GHG emissions 4% below its baseline (average 1998 to 2001 CO 2 emissions) by 2006. The oat hull project has produced results that exceed the CCX commitment. Figure 3 illustrates UI’s CO 2 emissions from 1998 through 2005 by fuel source. Burning coal accounts for the vast majority of UI’s CO 2 emissions. The impact of the oat hull project is clearly seen in the decreased CO 2 emissions from the CFB boiler.
3. UI’s CO2 emissions 1998 through 2005 by fuel type. Source: University of Iowa
Stoker Boiler Oat Hull Test Burn ResultsThe stoker boiler oat hull test burn commenced on September 6, 2006. Setup was completed by locking out the parts of the biomass system that would not be used for the test. The path of oat hulls from the silo was directed to the rear reinjector port in the back of the boiler, directly across from feeder #3. After starting the blower only, without any oat hulls, the boiler was allowed to adjust for the extra air being injected. After about 30 minutes of running air only, the oat hulls were injected into the boiler. When firing up to 1,000 lb/hr of oat hulls there was no dramatic change in furnace conditions. By just observing the firebox it was difficult to tell if any hulls were being injected into the boiler. Over the next three hours the rate of hull injection was raised to 3,000 lb/hr. At 3,000 lb/hr the test was stopped due to flame impingement on the front wall around feeder #3. Hulls and air coming from the reinjector port were pushing the flame across the boiler and up the front wall. Although it uncovered some problems, the test was considered successful because it answered two big questions:
With initial questions of safety, combustion control, and opacity implications answered, the power plant’s staff are now planning to test oat hull injection from a location on the furnace front wall. This should allow a substantially higher maximum oat hull co-firing rate and produce more controllable furnace condition. |