Regulatory agencies are becoming more and more critical of phosphorus-based water treatment chemicals because they can have a negative impact on lakes and waterways. In the past, treating cooling water with non-phosphorus products has not always kept systems in top condition, but recent testing has proven one new method works as advertised.
In recent years, public and regulatory bodies have heightened their protection efforts for our natural resources in order to protect against excessive water usage and harm to the environment. For good reason, it is no longer acceptable for industries to simply open up their valves and discharge unregulated waste streams to the environment. Companies, and even municipalities, are now subject to thorough review and comment periods to justify a facility’s proposed discharge rates and water composition.
In the realm of water treatment, one area that has historically come up during the environmental review cycle is the evaluation of additives used to treat cooling water systems. Chromate, a former chemical additive in cooling water treatment, was discovered to be carcinogenic and its use is now banned.
Phosphorus Usage
Since the early 1970s, the use of phosphorus-bearing compounds in industrial cooling water treatment programs has been commonplace. Phosphorus (P)—a common constituent of agricultural fertilizer, and municipal and industrial waste—is currently being reviewed by state agencies due to its impact on the eutrophication of lakes and other waterways.
Phosphorus has long been recognized as the controlling factor in plant and algae growth for many lakes and streams. A minor increase in phosphorus can fuel substantial increases in both aquatic plant and algae growth, which can have severe impacts on downstream bodies of water, such as rivers, lakes, or the Gulf of Mexico.
Phosphorus can originate from municipal and industrial facilities that discharge water, as well as from runoff from agricultural areas that makes its way into local water sources. Many parts of the country are regulating the acceptable P discharge levels well below those commonly employed in traditional cooling water treatments for scale and corrosion control. This represents a significant challenge for facilities operating under direct discharge permits.
Typical alkaline all-organic cooling water programs where phosphonates are present as scale inhibitors can have phosphorus levels from 0.3 ppm to 2.5 ppm, while stabilized phosphate programs can have phosphorus levels as high as 7.0 ppm. Alternative corrosion inhibitor options may not be viable because they are often based on metals such as zinc or molybdate, which are also being closely regulated in terms of acceptable discharge levels.
Without an effective corrosion and deposit (scale) control program in place, industrial-cooling systems could be compromised in a relatively short period of time. Facilities could experience significantly higher operating costs. For example, scale buildup on condenser tubes could affect vacuum and thereby reduce power generation.
Searching for Alternatives
Recognizing the challenges facing the industry, specialty chemical providers have been diligently working to find an economically viable solution that can deliver the protection required. Suppliers have made non-metallic corrosion inhibitors commercially available, but these organic inhibitors are often based on low molecular weight polymers, phosphonates, and amino phosphonates.
Some of these organic molecules have proven to be successful carbon steel corrosion inhibitors with improved environmental acceptability, but they may have some technical concerns. Organic molecules may be more susceptible to oxidizing biocides than inorganic molecules. Many azole-based copper corrosion inhibitors are susceptible to degradation by oxidizing biocides.
Hydroxyphosphonic acid has shown susceptibility to chlorine, even at low levels. This presents a problem because oxidizing biocides are a necessary part of any successful water treatment program for control of potentially harmful bacteria, such as Legionella. Organic inhibitors based on the phosphorus molecule do not meet the discharge requirement for P due to the reversion of some of the organic phosphonate to orthophosphate (O-PO4) and eventually to phosphorus. Unfortunately many alternative programs have failed to properly control the corrosion rates in systems without the use of P-bearing inhibitors.
Table 1 illustrates a “green chemistry” alternative evaluation conducted by U.S. Water Services Inc. (U.S. Water), a company based in St. Michael, Minn. The table contains information related to the various alternatives where the values are the subjective ratings determined by the authors of “The Pursuit of a Green Carbon Steel Corrosion Inhibitor: Part 1 of 4” published by Modern Pumping Today in January 2015 (4 is the best score and 1 is the worst). Based on this information, U.S. Water developed a unique cooling water treatment technology, called PhosZero, a zero-P cooling water chemistry.
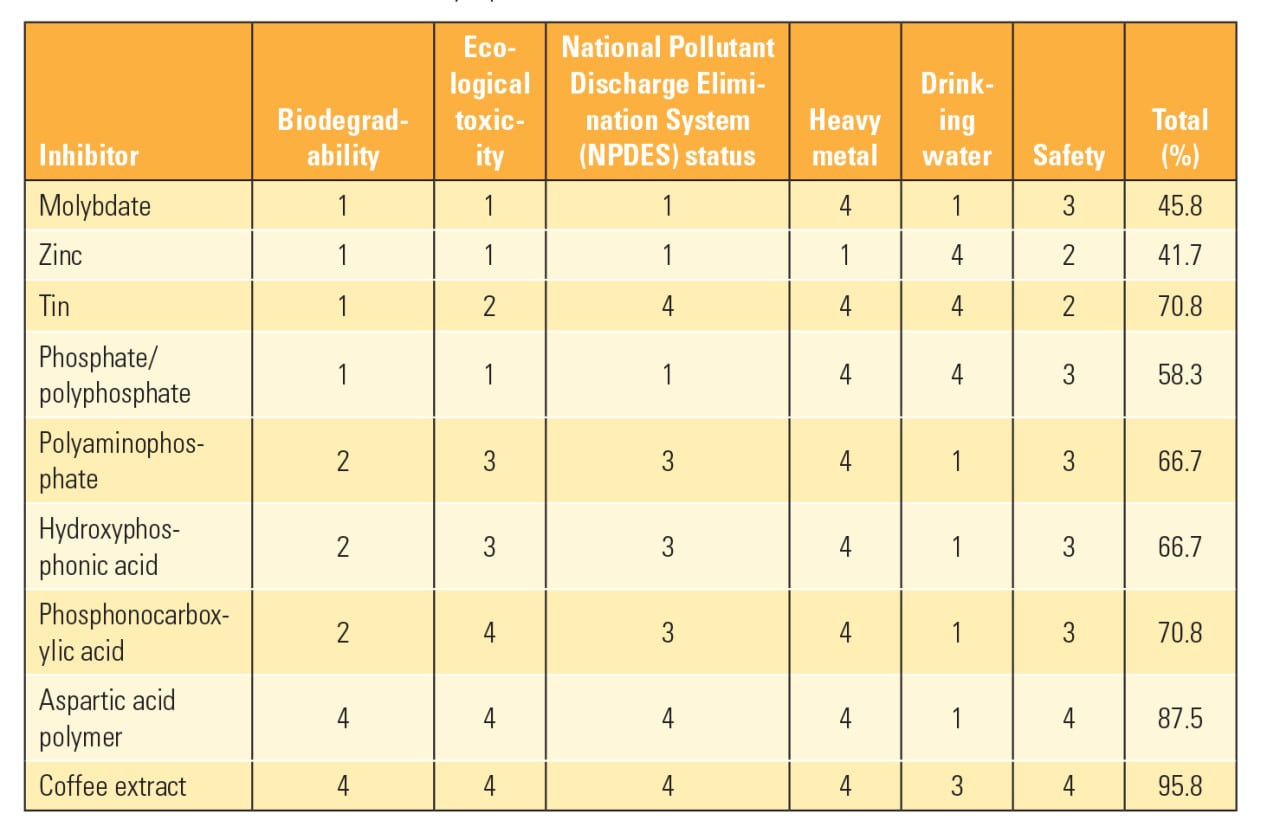 |
| Table 1. Water chemistry alternatives. This table shows the subjective ratings of various water treatment chemistry options. Source: U.S. Water |
Finding the Best Option
A Midwest refining plant reached out to U.S. Water for help achieving its goal of reducing P concentrations in its discharge. The plant had two different cooling towers due to different process conditions and both utilized induced-draft counter-flow cooling towers. Traditional water treatment programs were being utilized in the treatment of the cooling towers, but due to discharge regulations, the client needed to find a viable chemical treatment alternative. The new P-free program was implemented at the plant.
In order to ensure proper system protection, the corrosion rates were carefully monitored using corrosion coupons as well as an online corrator. The client wanted to maintain the same water efficiency, that is, not reduce the cycles of concentration, which yielded a Langelier Saturation Index (LSI) value of approximately 2.25. This LSI represents a relatively high scaling potential and the heat transfer surfaces were monitored closely as well. During the course of the entire trial, there was no observed accumulation of scale that would impede the heat transfer efficiencies. Table 2 lists the pertinent cooling water values during the trial.
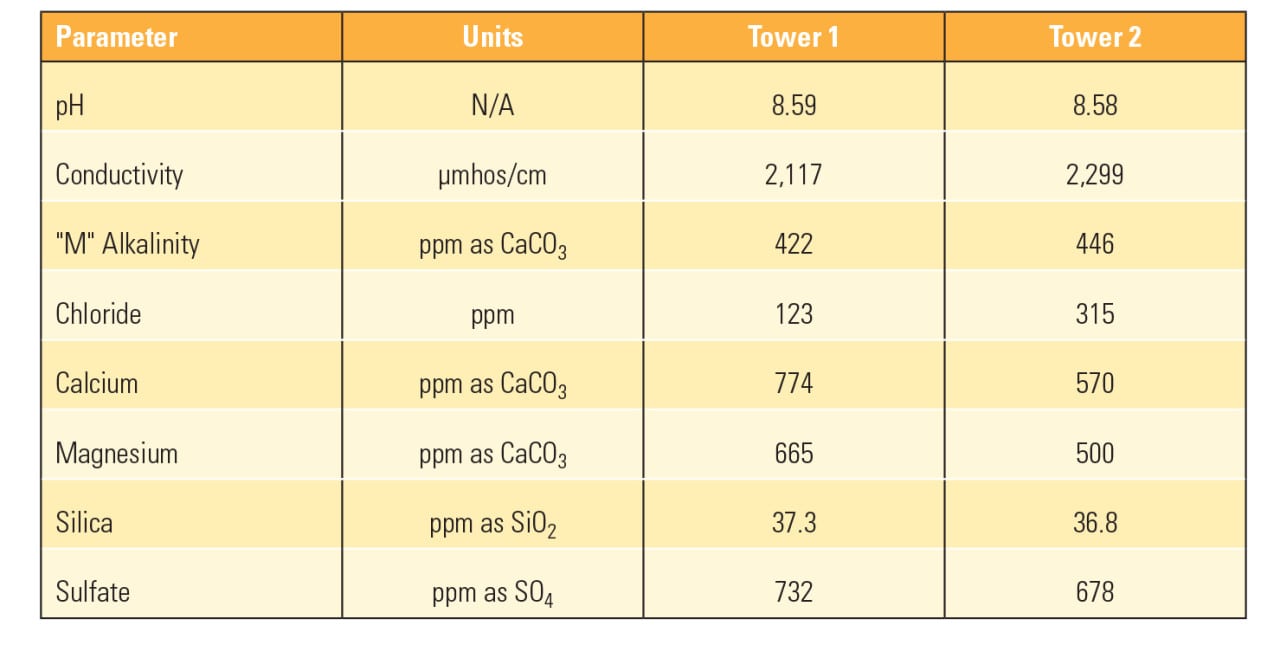 |
| Table 2. Phosphorus-free test results. The water treatment program at a Midwest refining plant was changed to a phosphorus-free chemistry with the results shown here. Source: U.S. Water |
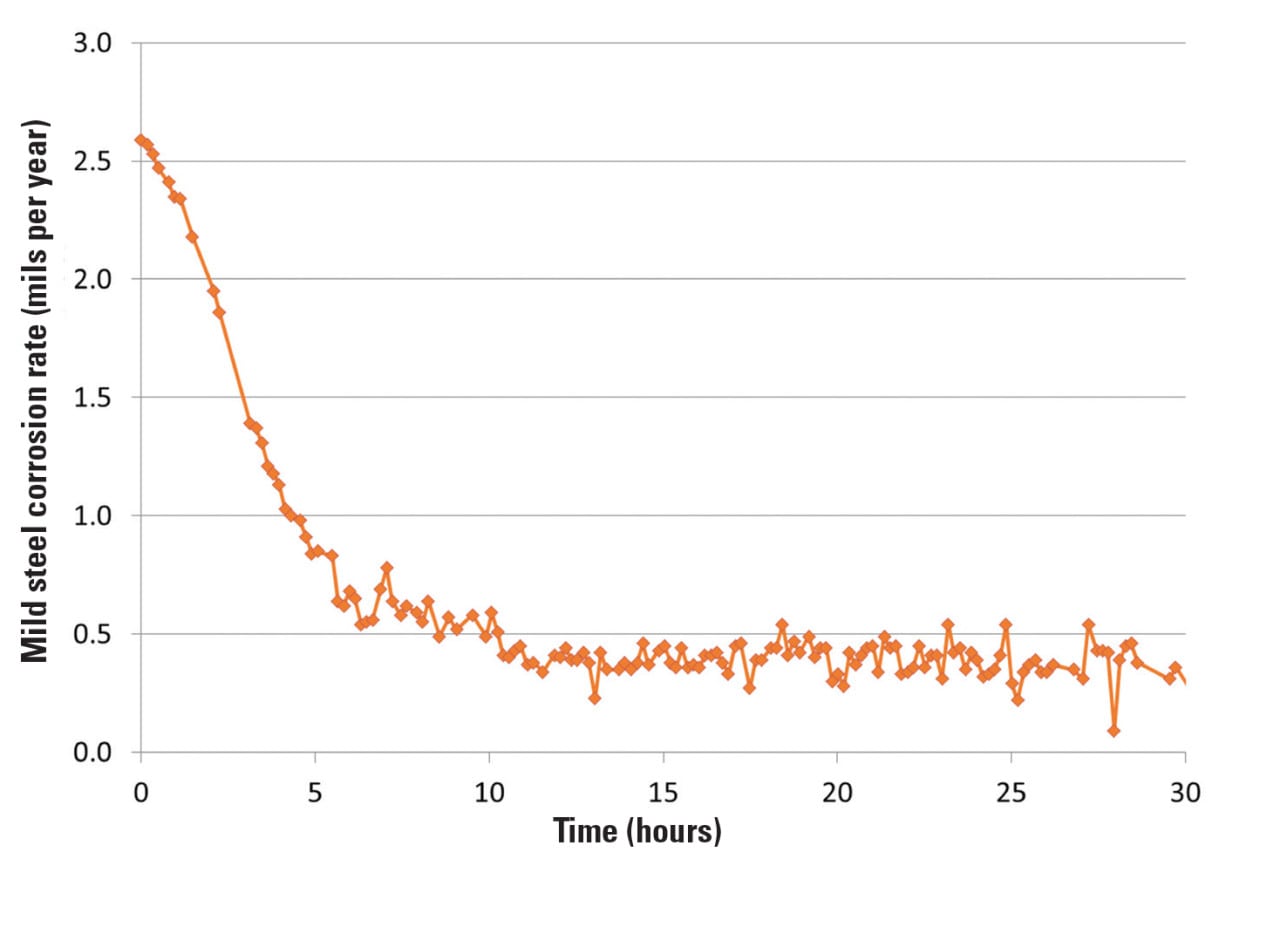
After the zero-P program was implemented, the corrosion rates improved drastically within the first five hours of application. The online corrator corrosion rates for the mild steel (Figure 1) showed the significant improvement during the implementation phase. The corrosion rate in mils per year (mpy) was reduced from greater than 2.5 mpy to less than 0.5 mpy.
In this study, corrosion coupons were employed in addition to the corrator to monitor the cumulative corrosion rates during a 45- to 60-day exposure period. The results are shown in Figure 2.
 |
| 2. Coupons tell the story. Corrosion coupons employed during a 45- to 60-day test were used to calculate corrosion rates, which ranged from 0.702 mils per year (mpy) to 1.661 mpy. Source: U.S. Water |
The results were better than anticipated and the primary objective of eliminating P from the cooling water chemical treatment program was attained. In addition, the zero-P treatment program provided a significant reduction in the corrosion rates. The corrosion rates dropped from an average of about 4 mpy to an average of around 1.3 mpy.
Another Effective PhosZero Implementation
In a second study, Minnesota Power’s Arrowhead High-Voltage Direct Current Terminal was experiencing scale and white rust buildup inside its cooling tower condenser tubes, creating obstacles cooling the inverter. If left untreated, the buildup could have resulted in a costly asset replacement and unscheduled downtime.
At the same time, the Minnesota Pollution Control Agency (MPCA) was instituting new phosphate discharge limits and encouraging plant management to meet the new state limits before implementation. Minnesota Power reached out to ALLETE sister company U.S. Water for assistance in achieving system efficiency and meeting new state discharge limits.
U.S. Water worked with Minnesota Power’s team to implement a zero-P program into its system. The PhosZero chemistry provided low enough toxicity to gain the approval of the MPCA, yet strong enough scale inhibition properties and corrosion protection to keep its system running efficiently.
A few months after introducing the zero-P chemistry into the system, hardness balance testing showed that scale was being removed. Figure 3 shows the success of the zero-P program with before and after results of mild metal and zinc corrosion coupons.
The results signify a paradigm shift in the water treatment industry. The norm used to be for facilities to have to settle for compromised results or additional water use when using alternate chemicals that didn’t contain P. Now the industry is able to meet low-P concentrations in its discharges while still protecting its critical assets. ■
—Mike Mowbray is director of product management for U.S. Water.