Given the costs and other concerns about currently available technologies for capturing and storing carbon dioxide from fossil-fueled power plants, interest in new technologies remains high. Here’s a look at some potentially promising approaches that are advancing the technology frontier.
Frontiers represent the boundary between the known and the unknown. As researchers attempt to push the frontier of carbon capture and storage (CCS) technology, they are encountering both tantalizing possibilities and daunting obstacles.
We know that carbon dioxide (CO2) from fossil-fueled power plants can be captured, but current options for doing so are expensive, present operational challenges for the rest of the plant, and are difficult to initiate and complete. (For more on current technologies, see “CCS Development, the Key to Coal Power’s Future, Is Slow” in this issue.) That’s why researchers are scouting out new approaches that are cheaper as well as easier to install and operate.
Although coal-fired plants are the initial target for CCS use, natural gas–fired plants may also be subject to future CO2 emissions limits, which is why the Electric Power Research Institute (EPRI) is studying the technical and economic issues associated with use of an advanced amine-based system at a natural gas combined cycle plant. Amine-based post-combustion capture processes are the most familiar. Some of the other schemes being explored for carbon capture look farther afield, as you’ll see.
Ultimately, as Dr. Larry Baxter noted at the February ARPA-E Summit, CCS isn’t just a coal technology. The Brigham Young University (BYU) professor noted that to reach global CO2 emissions reduction goals, the technology would need to be applied to gas production, cement kilns, refineries, and other stationary sources.
Intriguing Approaches
It seems that hardly a week goes by without a story in some publication about a novel approach to capturing carbon dioxide, from gells to cement to “buckeyballs” and other metal organic frameworks to various membranes. With such early-stage technology, it seems likely that the feasibility of different approaches may depend on site-specific circumstances—at least until a smaller subset of commercially proven options rises to the top.
Mention of a particular approach in this article should not be taken as an endorsement. That said, projects selected for early-stage funding by the Department of Energy’s (DOE’s) Advanced Research Projects Agency-Energy (ARPA-E)—based on a combination of technology promise and the strength of the research and development (R&D) team—may be some of the more promising. As of Mar. 20, the ARPA-E site listed 15 projects funded by its Innovative Materials and Processes for Advanced Carbon Capture Technologies (IMPACCT) program. Brief descriptions of three different approaches that were represented at this year’s Summit follow.
Metal-Organic Frameworks
A number of research groups are exploring metal-organic frameworks as an alternative to amine-based carbon adsorption systems. A description of the University of California, Berkeley project illustrates both the promise and challenge of this line of exploration.
This project, which has been awarded nearly $5 million from ARPA-E, is “developing a method for identifying the best metal organic frameworks for use in capturing CO2 from the flue gas of coal-fired power plants. Metal organic frameworks are porous, crystalline compounds that, based on their chemical structure, vary considerably in terms of their capacity to grab hold of passing CO2 molecules and their ability to withstand the harsh conditions found in the gas exhaust of coal-fired power plants. Owing primarily to their high tunability, metal organic frameworks can have an incredibly wide range of different chemical and physical properties, so identifying the best to use for CO2 capture and storage can be a difficult task.”
In a CCS panel discussion at the Summit, Dr. Steven Kaye, the project lead for the UC Berkeley Mosaic Materials Project, part of the Lawrence Berkeley National Laboratory (Berkeley Lab) Cyclotron Road program, said this project was moving toward demonstration at the National Carbon Capture Center. (Note that most CCS research projects are collaborative efforts with many “parent” companies, departments, and funders.)
As this article was being written, Berkeley Lab issued a release about some of the group’s research results being published in Nature (“Cooperative insertion of CO2 in diamine-appended metal-organic frameworks”). In brief: “By appending a diamine molecule to the sponge-like solid materials known as metal-organic-frameworks (MOFs), the researchers were able to more than triple the CO2-scrubbing capacity of the MOFs, while significantly reducing parasitic energy.”
The press release explains: “Consisting of a metal center surrounded by organic ‘linker’ molecules, MOFs form a highly porous three-dimensional crystal framework with an extraordinarily large internal surface area—a MOF the size of a sugar cube if unfolded and flattened would blanket a football field. By altering their composition, MOFs can be tailored to serve as highly effective storage vessels for capturing and containing carbon dioxide.”
Supersonic Carbon Capture
One of the most unexpected approaches being explored borrows from the well-established supersonic engine industry. At both the ARPA-E Summit and MIT Energy Conference in February, outgoing ARPA-E Director Cheryl Martin was very enthusiastic about the work being conducted by ATK, a company that received roughly $2.7 million in ARPA-E funds and whose research has moved on to the next stage under the auspices of the DOE’s National Energy Technology Laboratory (NETL).
As the ARPA-E site explains, “Researchers at ATK and ACENT Laboratories are developing a device that relies on aerospace wind-tunnel technologies to turn CO2 into a condensed solid for collection and capture. ATK’s design incorporates a special nozzle that converges and diverges to expand flue gas, thereby cooling it off and turning the CO2 into solid particles which are removed from the system by a cyclonic separator. This technology is mechanically simple, contains no moving parts and generates no chemical waste, making it inexpensive to construct and operate, readily scalable, and easily integrated into existing facilities. The increase in the cost to coal-fired power plants associated with introduction of this system would be 50% less than current technologies.” EPRI and The Ohio State University have also been project collaborators.
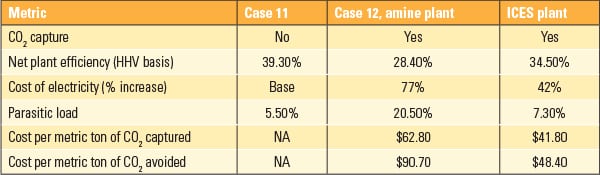
Martin noted that ATK’s approach, while attractive because it uses no chemicals and has few moving parts, still requires a fairly large footprint. A July 30, 2014, presentation to NETL on ATK’s bench scale project status update, shows the inertial CO2 extraction system (ICES) taking up about 8,000 m2—compared to 20,000 to 30,000 m2 for an amine plant of similar capacity, it says (Figure 1). It suggests that “ICES nozzle and compressor stacking can further reduce footprint by 30–40%.”
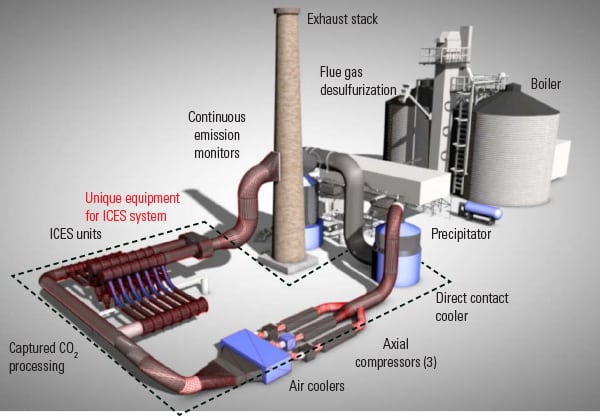 |
| 1. ICES mockup. This representation shows how an inertial CO2 extraction system (ICES) would be added to a coal-fired power plant. Courtesy: ATK |
At the Summit, Bon Calayag, a senior program manager for ATK Defense Group, noted that this project evolved because what was a problem for the parent company’s industry, aerospace and defense, looked like a solution for another industry, power generation. Supersonic condensation in wind tunnels is a real problem, as it can damage the articles under test; condensation of flue gas, however, allows extraction of the CO2. (For more details, see the ARPA-E video) ATK is looking for “adjacent uses” like hydrogen production for chemical companies to finance work on carbon capture.
Both the NETL presentation and one shared at the ARPA-E Summit referenced a 2013 preliminary techno-economic study by WorleyParsons that compared ICES very favorably to an amine-based CCS plant (Table 1). Dropping the parasitic load from 20.5% to 7.3% in itself would be a notable improvement.
Cryogenic Carbon Capture
Another “extreme” approach to carbon capture, which has received just over $5 million in ARPA-E-funding, is the trademarked Cryogenic Carbon Capture (CCC) developed by Sustainable Energy Solutions (SES). Funding went to a team comprising SES, American Air Liquide, BYU, and GE Global Research. BYU’s Dr. Baxter is cofounder and technical director of SES. Of the three projects mentioned in this article, the SES technology is farthest along the development track.
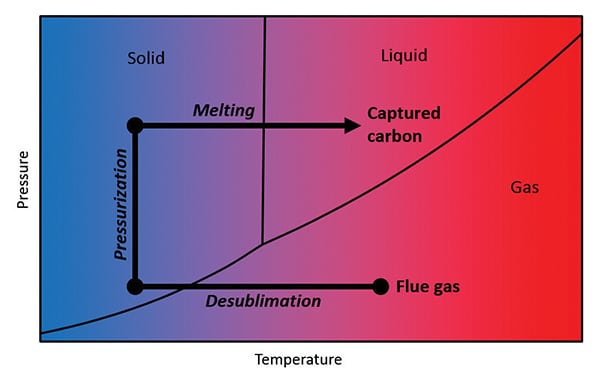
As the ARPA-E project site explains, “SES is developing a process to capture CO2 from the exhaust gas of coal-fired power plants by desublimation—the conversion of a gas to a solid. Capturing CO2 as a solid and delivering it as a liquid avoids the large energy cost of CO2 gas compression.” (For more details of the bench test stage, see the video at bit.ly/1I7R8ju.)
The company claims its post-combustion approach “has the potential to reduce carbon emissions from fossil-fueled power plants by 95–99%, at half the cost and energy of current state-of-the-art carbon capture processes.” As SES’s Kyler Stitt told me at the Summit, the system has been successfully tested in various demonstration projects, including, most recently, over several months at the Dave Johnston Power Plant near Casper, Wyo., with about 95% capture from a slipstream, on average.
The system takes the flue gas stream down to about –140C, which “freezes out” the CO2 plus other pollutants like SOx, NOx, and mercury, which are separated out (Figure 2). The company website explains that “The solid CO2 is separated from the remaining gas, pressurized, melted, and delivered at pipeline pressure.” The captured CO2 can be used in applications including enhanced oil recovery (EOR) and biofuels production. At low enough temperatures, the company says “the exhaust exiting the stack actually has less CO2 than the surrounding air.”
Stitt said SES modeling shows the process’s parasitic load can be brought down to about 14%. With additional improvements, SES hopes to bring that down further (Figure 3). The company says that in greenfield installations, “the pollutant capture capability of CCC can offset the cost of traditional pollutant removal systems.”
Addressing the Sequestration Challenge
Once CO2 is captured, it still has to be stored or “sequestered” safely and permanently in order to have any chance of having any positive effect on total global climate-forcing emissions. That’s not always easy.
Today, the main way to commercially store CO2 from power plants is to send it underground, in the service of EOR. In such cases, the CO2 has an economic value as an industrial process input. But not all geologies will be appropriate to the gas’s sequestration, with or without the option for EOR, so unfortunately sited emission sources would need to pipe the gas potentially very long distances. Add to that cost the vast unknowns regarding environmental unintended consequences and legal liabilities, and the prospects for non-EOR-based long-term underground storage look iffy. (To briefly mention just one of the potential environmental unknowns, EPRI is working on a report that looks at the impact of CO2 on groundwater. Given the concerns about the effects of fracking and fluid reinjection on groundwater, this is a nontrivial consideration.)
Fortunately, that’s not stopping researchers from exploring new options. SES, for example, says its patented technology can achieve stable storage in natural saline aquifers of “up to seven times more CO2 than traditional methods.”
Aside from underground burial of captured CO2, what other options are there? Several have been proposed and explored. The most likely alternatives involve some sort of encapsulation in a stable (and perhaps even commercially useful) solid. POWER has covered several of these projects over the years, including Skyonic Corp., whose October 2014–dedicated Capitol SkyMine near San Antonio, Texas, is designed to mineralize an adjacent cement plant’s CO2 emissions and sell them as baking soda, bleach, and hydrochloric acid.
Another enterprise, Calera Corp., converts CO2 into calcium and magnesium carbonates for use in manufacturing carbon-negative products. As of March this year, the company says its pilot plant in Moss Landing, Calif., has been in operation for approximately two years “and the calcium carbonate produced from the capture and conversion of CO2 from the neighboring gas power plant has been used in a variety of demonstration and commercial projects.”
Calera’s technology uses flue gas directly from the source, without concentrating or compressing it, and “has demonstrated the technology on CO2 from gas power plants and from coal burning. The flue gas containing CO2 is contacted in a scrubber with an aqueous alkalinity solution that effectively removes the CO2 and then contacted with a calcium source that results in the formation of the special calcium carbonate product that is then dried to a free flowing powder.” That powder has been used in products ranging from “board type products” to plant holders and benches.
Scale Matters
At both the ARPA-E and MIT events earlier this year, industry leaders expressed confidence that CCS is a realistic option for reducing greenhouse gas emissions. At the MIT event, for example, Dirk Smit, chief scientist for Shell Global, said Shell thinks CCS “is a very viable alternative” when “enabled by carbon pricing,” which the company is in favor of. For a sustainable energy system, we need “affordable carbon capture technology” but also carbon storage “at scale,” because affordability, he said, is a scale question.
Scale, many agree, poses the greater challenge. Though Shell is involved with three carbon capture “demonstration” projects (Gorgon in Australia, Mongstad in Norway, and Quest in Canada), Smit noted there is still “difficult work to be done.” Sequestering carbon at scale requires using stable reservoirs that may not always be available, which means that CCS (at least in its current form) may be relegated to “isolated projects.”
Speed Matters
Despite the welcome kickstart that ARPA-E and other funding sources provide to promising ideas, and despite the number of researchers working on improved carbon capture and sequestration schemes, under the current R&D paradigm, it could easily be a decade or more before any effective, efficient, and economical CCS approaches are sufficiently proven to be widely and swiftly adopted around the world.
For example, that supersonic ICES method is being “bench-scale” tested, prior to “potential future pilot-scale testing with coal-fired flue gas. Achieving this goal,” the NETL brief says, “will make progress toward meeting the DOE goal to demonstrate second-generation technologies that can capture 90 percent of the CO2 at less than $40 per tonne in the 2020–2025 timeframe.”
Will that be fast enough—especially considering that power generators dislike adopting brand-new technologies, and that developing nations around the world continue to build new fossil-fueled generation?
For those who support the continued use of coal and gas—whether because their jobs are tied to those fuels or because fossil fuels offer high energy density and dispatchable power—the ability to use off-the-shelf CCS technology is likely to offer a far more secure way to ensure a bright future for fossil fuels globally than the current political and legal tactics.
Perhaps what CCS needs is the pedal-to-the-metal focus and resources of an Elon Musk. ■
— Gail Reitenbach, PhD is editor of POWER.