New air emission rules limit the amount of mercury (Hg) air emissions from coal-fired power plants. Many plant owners may find leveraging the Hg removal co-benefits available from SCR Hg oxidation and FGD Hg collection a more attractive option than constructing new equipment or using other expensive mitigation technologies.
The new Mercury and Air Toxics Standards (MATS) have given coal-fired power plant owners a short time to bring their plants into compliance with rules that require significantly less mercury (Hg) in stack gases emitted into the atmosphere. MATS applies to U.S. plants, but the move to reduce power plant mercury emissions has gone global. In January 2013, after four years of negotiations, 140 nations signed the first legally binding agreement to control mercury emissions from power plants (and many other sources and products). The official signing of the Minamata Convention (named after a city in Japan that experienced environmental damage caused by industrial mercury discharges into local rivers) is scheduled for October of this year. New rules are expected to go into force over the next three to five years, with full effect by 2020.
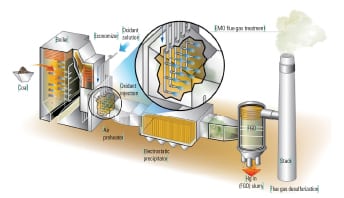
Flue gas from coal-fired power plants may contain Hg in three different species or forms. First, the Hg may be in particulate form (HgP), which is typically removed with the ash and unburned carbon in an electrostatic precipitator or fabric filter. Second, the Hg may be in oxidized form (Hg2+), which is water-soluble and can be removed by a wet flue gas desulfurization system (FGD). The Hg may also appear in its elemental form (Hg0), which is not water-soluble and usually passes unchanged straight through most air quality management systems and into the atmosphere.
The potential synergy from combining SCR and FGD technologies for mercury removal has been known for some time. Data collected during the Environmental Protection Agency’s (EPA’s) Information Collection Request campaigns in 1998, 2005, and updated in 2009, showed that mercury capture in bituminous-fired units with cold-side electrostatic precipitators increased from about 36% to 75% when a wet FGD was also in service. The data also showed that the amount of Hg removal increased to >95% with the SCR in-service compared to when it was in bypass. More recent studies have produced similar results (see “Determining AQCS Mercury Removal Co-Benefits,” in POWER’ s July 2010 issue and “An SCR Can Provide Mercury Removal Co-Benefits,” October 2011).
You Have Options
The two most familiar options to reduce mercury emissions are a high capital cost retrofit with equipment specifically designed for mercury removal (such as TOXECON) or using an expensive combustion gas additive, such as activated carbon injection (ACI), that will adsorb the mercury for capture in a downstream filtration system. The effectiveness of both approaches is highly dependent on the type of fuel consumed by the plant.
A third option, the use of existing equipment, or “co-benefit option,” can be the most cost effective: leverage air quality control equipment already in place at most coal-fired plants to produce the necessary Hg reduction without an extended plant outage required for an equipment retrofit. This two-step approach occurs by first oxidizing the Hg0 present in the stack gas as it passes through the SCR and then removing the Hg2+ in the FGD. In many cases, the catalyst used in the SCR must be optimized for site-specific conditions to achieve the desired Hg0 oxidation rate and therefore the overall mercury removal efficiency. In addition, recent advancements in FGD technology and additives help to prevent re-emission of the captured oxidized mercury. Another option is to utilize supplemental mercury capture using sorbents such as ACI in combination with the existing equipment. This option can be used to increase mercury removal efficiency where specific plant equipment and/or conditions do not allow optimization of the SCR/FGD co-benefit.
Cormetech recently commercialized its Oxidized Mercury Emissions Technology (COMET) that can be used to characterize reactor performance for a set of plant-specific features and fuels, determine the correct catalyst formulation so that NOx removal rates remain unchanged, and provide a specific catalyst management plan to obtain the required levels of mercury oxidation through the SCR. The potential mercury removal co-benefits and cost savings can be substantial.
SCR Hg Oxidation Co-Benefit
The SCR of NOx using NH3 as the reductant for V2O5 – (WO3 or MO3)/TiO2 catalysts is the current best available control technology for NOx emissions from coal-fired utility boilers. SCR has demonstrated NOx removal efficiencies of 90% or more. The primary NOx reduction reaction proceeds according to the stoichiometry described by the following two equations (note that NOx levels in the flue gases from coal-fired boilers typically contain >90% NO).
Equation 1: 4NO + 4NH3 + O2 4N2 + 6H2O
Equation 2: 2NO + 2NO2 + 4NH3 4N2 + 6H2O
SCR catalysts are also active for the oxidation of Hg0 by chlorine as HCl and/or by bromine as HBr, as described by Equation 3 for HCl. The conversion of Hg0, which is water-insoluble, to HgCl2 or HgBr2 (water-soluble oxidized mercury Hg2+) allows for capture of mercury in a downstream flue gas desulfurization system.
Equation 3: 2Hg0 + 4HCl + O2 2HgCl2 + 4H2O
SCR Does Double Duty
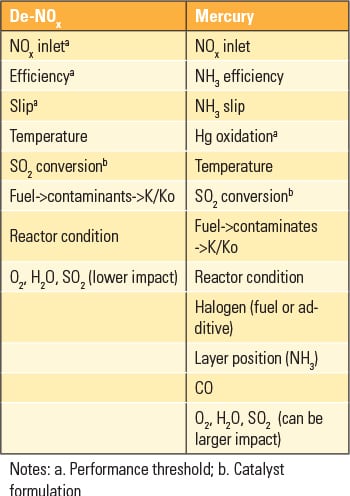
Differences between NOx and Hg removal performance must be considered within and surrounding the SCR. From an SCR perspective, de-NOx performance is well defined and controlled within the SCR reactor by catalyst selection, cross-sectional area, number of modules, and so on. However, total Hg removal must carefully consider the mercury removal performance of equipment downstream of the SCR, such as the air heater, particulate control device(s), and the FGD. A number of system-level factors relative to characterizing and understanding catalyst performance must also be considered (Table 1).
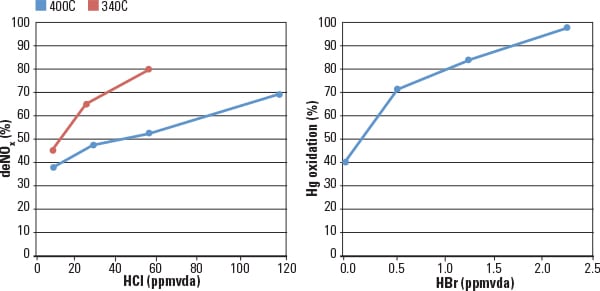
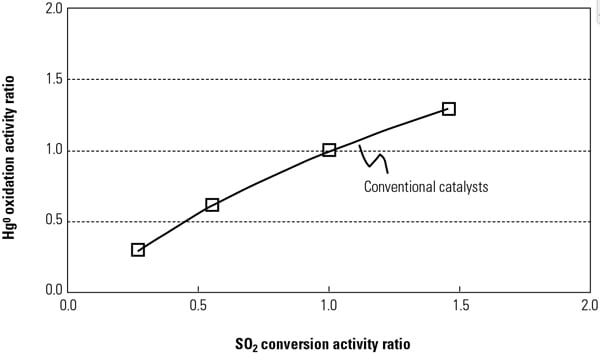
Cormetech’s early product development work to quantify the co-benefits SCR + FGD mercury removal used a mercury activity test reactor system in conjunction with a multi-layer catalyst system to perform multiple parametric tests. For example, one set of tests characterized layer position and halogen content for a given catalyst. Tests were also conducted on new as well as aged catalysts taken from operating plants. This critical data enables accurate prediction of catalyst Hg oxidation performance for unique applications. Typical results from those tests show that NOx reduction is a strong function of the operating temperature of the reactor and the HCl present in the gas. In addition, the Hg0 oxidation to Hg2+ is a strong function of the halogen content (HBr) and operating temperature (Figure 1).
Catalyst Design and Selection
Traditional catalyst management techniques provide very accurate predictions of de-NOx performance for various fuels and fuel additives. At the heart of the catalyst management process for de-NOx is a simplified method to describe catalyst oxidation potential, shown in Figure 1 and represented by Equation 4.
Equation 4: K de-NOx/AV = ln (1– de-NOx efficiency), where K de-NOx = catalyst de-NOx activity, AV = area velocity, α = NH3:NOx molar ratio = 1
The addition of mercury oxidation as a performance feature of the SCR adds complexity to the management process where both de-NOx and Hg oxidation needs must be managed simultaneously. Thus it is important to develop equations and tools to accurately model mercury oxidation performance as has been done with de-NOx. Again, a simplified approach to describe catalyst oxidation potential is shown in Figure 1 and represented by Equation 5.
Equation 5: K HgOx/AV = ln (1– Hg0 oxidation) at α = variable, where K HgOx = catalyst Hg oxidation activity and AV = area velocity
Although the equations are similar, the usefulness of the equation for Hg oxidation is influenced to a greater extent by the ammonia concentration. Therefore, instead of simplifying the assessment to one molar ratio, multiple molar ratios must be considered to properly assess the performance capability of a given catalyst layer in a particular position within the reactor. Plant-specific conditions related to fuel parameters, such as halogen and Hg concentration, also need to be established in the same way as the traditional inputs for de-NOx, such as temperature, inlet NOx, O2, H2O, SO2, and SO3.
The optimal SCR catalyst design for a given unit will maximize the rates of the de-NOx (Equations 1 and 2) and Hg oxidation (Equation 3) reactions, while minimizing the rate of the SO2 oxidation reaction, SO2 +½O2 SO3.
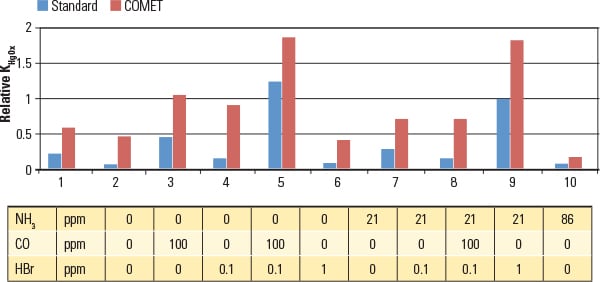
Routinely, SCR catalyst is formulated to a maximum allowed SO2 oxidation rate to minimize the negative effects of high SO3 caused by air preheater plugging or opacity concerns (visible plume), while continuing to meet the de-NOx and NH3 slip requirements. In other words, the de-NOx reduction is limited by the expected SO2 oxidation. COMET is designed to function within these de-NOx operational constraints without impacting its mercury reduction function. Other factors impact the catalyst design, such as Hg0 oxidation that occurs across the air preheater, the efficiency of HgP removal, the Hg2+ capture efficiency in the FGD, HCl and HBr concentration in the flue gas, SCR operating temperatures (particularly for cycling and load-following units), and so on. Figure 2 illustrates improvements that have been made in Hg oxidation performance as a function of key input parameters, specifically NH3, CO, and halogen content, while maintaining constant de-NOx and SO2 conversion rates.
Managing Catalyst Performance
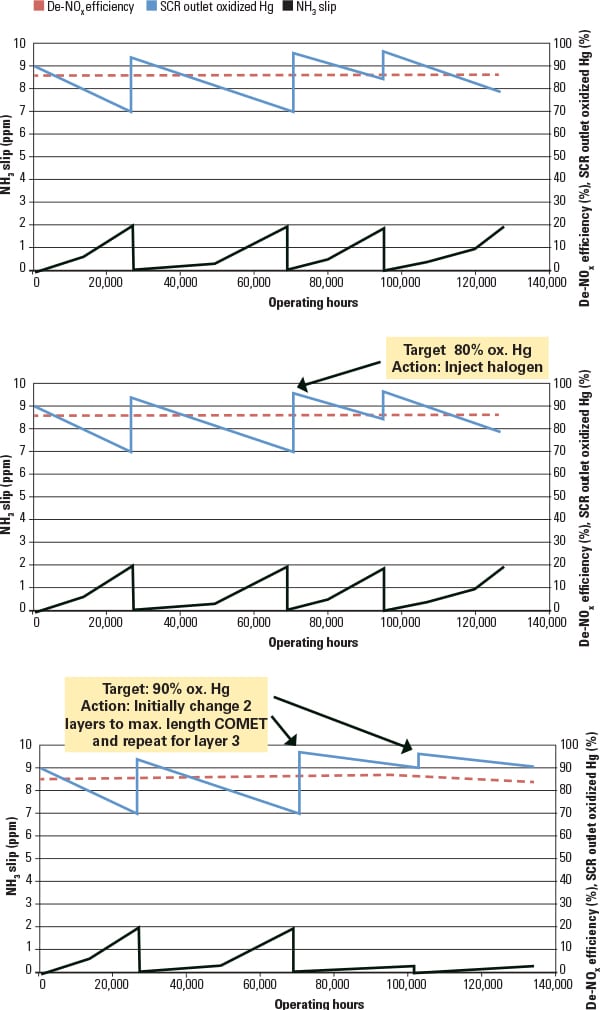
Once SCR de-NOx and Hg0 oxidation targets are established, catalyst management options can be explored. Figure 3 presents a series of catalyst management strategies over the lifetime of a catalyst. Figure 3, top, illustrates a typical baseline catalyst management plan with a de-NOx rate of 85% and an Hg0 oxidation rate of 70%. If at some time in the future the desired Hg0 oxidation rate were increased from 70% to 80% at 70,000 hours of operation, then the catalyst management plan would be modified to increase the halogen injection (Figure 3, middle). If the required removal rate were to increase from 70% to 90% at 70,000 hour of operation then a catalyst change would be required (Figure 3, bottom). Note that combinations of additives and catalyst design options would also be considered when updating the catalyst management plan to achieve the most cost-effective system or to add additional operating flexibility to the unit.
Figure 3 illustrates a single catalyst management plan responding to a hypothetical change in the required mercury removal rate in the future. There are many other combinations available than just those illustrated. The SCR can be tuned should Hg removal rates be ratcheted up again in the future or if there are other operating changes in the plant, such as a fuel switch.
More aggressive emissions rules are likely in the future, so the best upgrade path for many plants is to keep their future options open and capital costs low. COMET is a highly flexible, cost-effective compliance strategy for a unit, plant, or even an entire fleet of coal-fired facilities. ■
— Chris Bertole, PhD (bertolecj@cormetech.com) is catalyst development manager and Scot Pritchard (pritchardsg@cormetech.com) is senior vice president, sales engineering for Cormetech Inc.