ORP as a Predictor of WFGD Chemistry and Wastewater Treatment
Recent studies have shown that system oxidation-reduction potential (ORP) is not only an important factor for predicting wet flue gas desulfurization (WFGD) absorber chemistry but also may be a predictor of process equipment corrosion and wastewater treatment requirements.
Purge streams of wet flue gas desulfurization (WFGD) units, which are one byproduct of controlling SO2 emissions from coal combustion, are being increasingly subjected to stricter wastewater regulations. Consequently, coal-fired power generators need a method for controlling the operational chemistry of these WFGD units. Upon implementation of a suitable control method, WFGD bleed stream chemistry and flow rate may be optimized, thereby resulting in improved performance of one or more downstream unit operations. A further benefit is reduced reagent and additive costs in various devicies, including the WFGD unit.
One control parameter of interest is the oxidation-reduction potential (ORP) of the bleed stream. Much like pH, the measurement of ORP can be taken in real time and integrated with other plant-monitoring data. By incorporating ORP measurements into a process control scheme for limestone forced-oxidized WFGD absorbers—along with various other control variables such as SO2 removal, absorber pH, reagent flow rate and/or one or more reaction stoichiometries, and/or gypsum purity—generators are able to manage the oxidation states of various dissolved metals in the slurry and the potential reemission of mercury. (Also see “How to Measure Corrosion Processes Faster and More Accurately,” May 2009 in the POWER archives and “Mercury Control: Capturing Mercury in Wet Scrubbers, Parts I and II,” July and September 2007, respectively, in the COAL POWER archives—both available at powermag.com.)
A further benefit is control of the corrosion rate of the absorber recirculation tank (ART) and other alloy parts within the system. Many utilities have had ORP excursion events in WFGD wastewater discharge where the ORP readings changed from 150 millivolts (mV) to 300 mV to a reading above 500 mV. Previously, these fluctuations have gone largely unexplained. We have determined that this magnitude of change in ORP, in an ART, due to coal composition and upstream air quality control system (AQCS) effects on WFGD absorber chemistry, can accelerate corrosion.
One potential solution to fluctuating ORP readings is to use integrated process controls designed to tune the upstream operation of the AQCS train to produce consistent inlet flow parameters to the WFGD tower, rather than operating each as an independent process. The control of the ORP level in a WFGD system may produce improved plant operations by reducing the amount of wastewater treatment necessary and helping mitigate mercury reemission.
Fundamentals of ORP
ORP is a measure of the potential for a chemical species either to acquire or release electrons. The potential is commonly measured by an ORP probe in units of millivolts, which can be measured in real time under online plant process conditions. Positive readings are indicative of a system operating in oxidizing conditions; negative readings indicate a system operating in reducing conditions. If a material comes into contact with a solution that has a higher oxidative potential, then a chemical reaction may occur in which the solution is reduced and the material is oxidized.
The ORP of WFGD slurry and effluent is driven by the presence or absence of strong oxidizers. Many WFGD units operate at a moderate ORP range of about 100 mV to 300 mV, thereby achieving, or yielding, a rather stable voltage reading over time. The range of 100 mV to 300 mV is referred to as “low” ORP in this article. Such WFGD units often have oxidizer concentrations within the slurry below 200 ppm. Other WFGD units operate at higher ORP values, often above 500 mV. Slurries with high ORP almost always contain a high concentration of at least one strong oxidizer such as persulfate (S2O8-2), peroxymonosulfate (HSO5–), or hypochlorite (OCl–).
Persulfate has been identified and quantified using ion chromatography on absorber slurry samples collected at several sites. This anion is the most powerful oxidant of the peroxygen family of compounds, and it becomes a more effective oxidizer at scrubber process temperatures above about 120F due to free radical formation. In several instances, strong oxidizers were measured at total residual concentrations over 1,000 ppm in WFGD absorber slurry samples exhibiting high ORP after the samples were removed from the system and analyzed in the laboratory. Operating WFGD units are observed to swing from one process condition to the other (high to low ORP), but few, if any, hold at an intermediate value for an extended period of time. The rate and magnitude of these changes in slurry chemistry are indicative of upstream process changes affecting absorber chemistry.
Once you determine the WFGD slurry ORP, you can predict the dominant oxidation state for the various constituents that may be present in the absorber slurry. For many metals, solubility is a function of the oxidation state. Therefore, once the ORP of a solution has been determined, a prediction can be made of the preferred oxidation state for a given chemical species in a solution. The predominant species of various metals, and other compounds or ions, can thus be determined. Using this knowledge, the ORP in WFGD slurry can then be controlled in order to control the speciation of various metal ions, as well as other compounds and ions.
The range of potential electrochemical states of a given material can be found within a Pourbaix diagram for a given chemical species and presented as a function of pH and electrochemical potential versus pH. For example, a variety of general predictions about the various phases of mercury, selenium, and manganese based upon ORP and pH can be made (see the table).
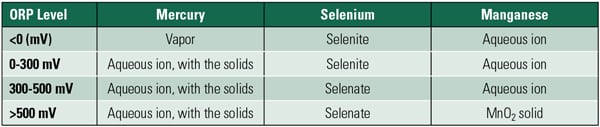 |
| Dominant forms of mercury, selenium, and manganese for approximate ORP levels. Source: Babcock & Wilcox Power Generation Group Inc. |
Dynamic Chemical Processes
By monitoring the ORP of WFGD absorber process slurry, changes within the system process and chemistry can be determined. Estimations of various parameters of WFGD absorber chemistry can also be determined, including the dominant oxidation states and phases of metals within the slurry, the potential reemission of mercury, and the risk of accelerated corrosion of the alloy vessel. Measurement of the system ORP also alerts operators to the likelihood of problems in downstream wastewater treatment (WWT) systems, especially the ratio and concentration of selenate ions to selenite ions within an effluent stream.
The chemistry inside a WFGD absorber is a complex system of hundreds of potentially changing ionic species and distinct compounds existing simultaneously throughout the slurry. The modeling of WFGD absorber slurries has been based on equilibrium thermodynamics to date. However, we have determined that in operating units, WFGD absorber slurry chemistry is more likely to be kinetically controlled. Most absorbers are operating at an unsteady state. Due to kinetic interaction, mercury could become reduced to the elemental state, thereby becoming vaporous and exiting the system boundaries in what is called mercury re-emission.
Another important aspect of ORP with relation to WFGD process chemistry is the reaction of any of the one or more strong oxidizers present with one or more halide ions present in solution, thereby resulting in increased demand for reagent in the absorber and a possible lowering of pH in the purge stream after the solids have been removed. Because persulfate is a powerful oxidant, it has the ability to convert some halide anions to their respective elemental state. Specifically, some chloride may convert to chlorine under high-ORP conditions:
Similar reactions occur between the oxidizer(s) and other halide species present (for example, bromide, iodide, and the like). Thus, the concentration of chlorine in absorber slurry is present as three species in equilibrium within the aqueous phase: dissolved gas (Cl2), hypochlorous acid, and ionic hypochlorite:
Within the typical WFGD operating range, equilibrium favors HOCl. The formation of H+ ions associated with this chemical reaction will cause a decrease in pH as halogen-containing species are liberated from the scrubber. In WFGD systems that are operating in pH control mode, the reagent feed controls will respond to the lower pH by adding more reagent. In systems with high ORP levels, gypsum formation may occur without the addition of oxidation air when sulfite reacts with strong oxygen containing oxidizing agents. Higher gypsum purity can result as excess limestone, normally an impurity within the gypsum product, is reacted to buffer the system from dropping pH.
Due to the electro-reactive nature of mercury, ORP levels are a main driver in controlling reemission, dissolution into the slurry, and solid phase retention. Higher ORP values in the ART are favored in order to maintain dissolved mercury. ORP levels above 500 mV often favor an increase in the dissolved mercury, with constant total mercury content in the slurry. A decrease in the ORP in an ART is an indication of a less-oxidizing environment, leading to elemental mercury formation and potentially release (or reemission). A possible chemical pathway for mercury reemission to occur is shown in Equation 3:
Swings in ORP value may also cause mercury to enter the elemental state and be reemitted from a WFGD tower.
Staying in Compliance
WWT systems are tuned to control metals present in influents and to produce effluents within a certain concentration range. Changes in the influx of these metals to WWT may disrupt performance if controls and operational parameters cannot respond quickly. As swings in WFGD and/or WWT process occur, ORP may ultimately produce changes in the dominant state of regulated metals, thus permitting the WFGD effluent flow rate to affect the mass flux of each species. In this situation, detrimental fluctuations in ORP may result in minimal removal of some metals and potentially result in out-of-compliance operation.
Controlling process ORP can lead to improved efficiency of WWT systems related to selenium removal. WFGD effluent ORP controls the precipitation of many regulated metals, particularly selenium. At low ORP, selenium exists mainly as selenite (SeO3-2) and can be removed by many WWT methods including chemical precipitation. At higher ORP levels (greater than about 300 mV,) selenium will predominantly occur as selenate (SeO4-2), which passes through many WWT systems.
The combined effects of over 1,000 ppm of total oxidizers and low pH potentially associated with high-ORP WFGD effluent streams can result in damage to bioreactor stock and/or increased reagent costs in WWT systems. Strong oxidizers present within WFGD effluent have the potential to upset biological processes. During high-ORP conditions, the WFGD would essentially be feeding bleach (hypochlorite), peroxide, and stronger oxidizers to downstream systems; such oxidizers can damage microbial health when fed to biological treatment units.
Furthermore, high ORP in a WFGD effluent can result in low pH of WWT influent. As described earlier, oxidizers will continue to react with halide ions in solution, thereby liberating a hydronium ion and thus lowering pH. When excess carbonate is available, it may buffer such impacts. Once unreacted limestone is removed from the slurry filtrate during dewatering, the pH buffering capacity of the system rapidly decreases while oxidizer and halide ion concentrations remain and are fed to WWT.
Materials Must Resist Corrosion
Materials coming into contact with WFGD slurry and effluent should be selected with careful consideration to the corrosive potential of high-ORP slurries containing ionic manganese. Strong oxidative content is present in high-ORP slurry leaving the scrubber, and very low pH levels may be seen downstream in conjunction with high ORP. While industry focus has been given to WFGD ART corrosion, the potential exists for similar corrosion to occur in process pumps, dewatering operations, vacuum systems for gypsum production, as well as in process piping and WWT equipment.
Alloy 2205 duplex stainless steel (UNS S32205) is proving susceptible to accelerated corrosion from slurries that contain precipitated, or non-solubilized, manganese species and/or high ORP levels. Within the WFGD absorber, the majority of the corrosive attack is observed below the slurry level in the ART. Under high-ORP conditions, the rate of corrosion can accelerate. When the ORP of operating WFGD slurries is pushed above about 500 mV, manganese normally soluble as ionic Mn+2 will oxidize and precipitate out as MnO2. When this precipitate contacts metal as part of a deposit, it serves as a galvanic cathode to exacerbate the fluoride and chloride driven under-deposit corrosion mechanism. Corrosion thus accelerated by manganese precipitation can be rapid and severe.
In many applications ceramic tile and linings are considered as alternatives to alloy. Wall-papering with UNS N10276 (Hastelloy C-276) has been performed in some installations. Some plastics and resins may also be susceptible to attack from high ORP levels. Strong oxidizers within the slurry effluent may react with and thereby degrade some polymer bonds, because high-ORP filtrate samples were observed to weaken and discolor HDPE bottles in a laboratory setting. Within substances such as fiberglass-reinforced plastic, such a reaction with resins could potentially cause some dissolution of the resin and/or lead to fiber delamination.
Plantwide Impact
The WFGD ART ORP and the WWT influent process ORP both need to be controlled. Currently, studies are being performed to define techniques to maintain steady ORP and to allow for greater fuel flexibility. Such control would afford utilities the option to obtain the most cost-effective fuel, maintain a constant effluent for wastewater, and provide a better treatment scheme. Coal yard and boiler operators will need to work in conjunction with the plant’s continuous emission monitoring systems, the WFGD system, and WWT plant operators to implement improvements that integrate the entire process. Learning how a parameter change upstream affects the WFGD and WWT systems is quickly becoming crucial. With full control of the AQCS process train and dewatering systems, control of WWT influent is expected.
The effects of blending coals, staging combustion, and swinging load can create issues with AQCS equipment, especially in environments where WFGD units are employed. The WFGD system serves as the catch basin for all flue gas byproducts as well as any fine ash not captured in an electrostatic precipitator or pulse jet fabric filter. Undesired ORP levels, or undesired fluctuations therein, can cause problems ranging from mercury reemission to increased corrosion and improper treatment of WFGD effluent. Adjustments to combustion processes may affect operating parameters of the WFGD environment as well as the WWT systems. Combustion systems need tuning to allow for efficient power generation, compliance with existing regulations, and flexibility to comply with anticipated, tighter water and solid discharge regulations.
Due to the potential for aggressive or accelerated corrosion of some alloy material in high-ORP environments, care should be taken on selection of alloys or materials in the WFGD slurry and filtrate contact zones. Constituents of the WFGD slurry will include limestone, gypsum, halide ions, and metals from the burned coal as well as silica from the ash and other up-stream constituents. The ORP levels in the tank can cause the metals to undergo phase partitioning or to change their solubility due to changes in their oxidation state.
— S.R. Brown ([email protected]) is AQCS engineer, R.F. DeVault ([email protected]) is research chemist, and D.B. Johnson ([email protected]) is field service engineer for Babcock & Wilcox Power Generation Group Inc.