Understanding Selective Catalytic Reduction Systems and SCR Design Considerations
Selective catalytic reduction (SCR) is an important emissions control technology utilized at many coal, biomass, waste-to-energy, and gas-fired power plants. Many items must be considered when designing SCR systems to optimize performance.
Without adequate emissions control systems, a significant amount of airborne NOx can be released from fuel combustion at power plants. NOx refers to nitric oxide (NO), nitrogen dioxide (NO2), and other oxides of nitrogen. There are a number of adverse environmental effects of releasing too much NOx into the atmosphere. The National Energy Technology Laboratory (NETL) lists the following:
■ NOx is a main constituent in the formation of ground-level ozone, which causes severe respiratory problems.
■ Respiratory problems may result from exposure to NO2 by itself, but also of concern is NOx reacting to form airborne nitrate particles or acid aerosols, which have similar effects.
■ Along with sulfur oxides (SOx), NOx contributes to the formation of acid rain and causes a wide range of environmental concerns.
■ NOx can deteriorate water quality by overloading the water with nutrients, causing an overabundance of algae.
■ Atmospheric nitrogen-containing particles decrease visibility.
■ NOx can react to form nitrous oxide (N2O), which is a greenhouse gas and contributes to global warming.
Selective Catalytic Reduction (SCR)
Selective catalytic reduction (SCR) systems remove NOx from flue gas emitted by power plant boilers, gas turbines, and other combustion sources. SCR selectively reduces NOx emissions by injecting ammonia (NH3) into the exhaust gas upstream of a catalyst. The NOx reacts with NH3 and oxygen (O2) to form nitrogen (N2) and water (H2O), primarily according to the following equations:
4NH3 + 4NO + O2 ➝ 4N2 + 6H2O
2NH3 + NO + NO2 ➝ 2N2 + 3H2O
4NH3 + 2NO2 + O2 ➝ 3N2 + 6H2O
The catalyst’s active surface is usually a noble metal, base metal (titanium or vanadium) oxide, or a zeolite-based material. Metal-based catalysts are typically applied as a coating over a metal or ceramic substrate, according to NETL, while zeolite catalysts are typically a homogeneous material that forms both the active surface and the substrate.
The geometric configuration of the catalyst body is designed for maximum surface area and minimum obstruction of the flue gas flow path to maximize conversion efficiency and minimize backpressure on the turbine. The most common configuration is a monolith, “honeycomb” design.
An important factor that affects the performance of SCR is the operating temperature. Base-metal catalysts have an operating temperature window for clean fuel applications of approximately 400F to 800F (~ 200C to 425C). The upper range of this temperature window can be increased using a zeolite catalyst to a maximum of 1,100F (~ 600C). Due to the required operating temperature range for conventional SCR catalyst (600F to 750F), integration into a heat recovery steam generator (HRSG) normally requires splitting of the high-pressure (HP) evaporator (or boiler) section to accommodate the SCR catalyst bed and ammonia injection equipment.
Selective Noncatalytic Reduction (SNCR)
Selective noncatalytic reduction (SNCR) is another post-combustion emissions control technology for reducing NOx. SNCR works by injecting an ammonia-based reagent into the furnace at a properly determined location. This technology is attractive because it requires a relatively low capital expense for installation.
More than half of utility boilers with SNCR are relatively small (200 MW or less), but about 24% are larger than 300 MW, according to the U.S. Environmental Protection Agency (EPA). More than 70% of the utility boilers using SNCR burn coal as the primary fuel and most of the others burn biomass. SNCR can be applied as a standalone NOx control or with other technologies such as combustion controls. SNCR systems can also be designed for seasonal operation in some cases, rather than operating them year-round.
“The SNCR usually operates at temperatures between 900C and 1,000C. Similar to the SCR, the nitrogen oxides are reduced to nitrogen by the injection of ammonia or urea. The correct adjustment of temperature defines the efficiency of the SNCR, as well as undesired side effects like ammonia slip,” explained Carsten Walddörfer, team leader for Research and Development (R&D), Product Management Clean Technology Systems, with Dürr Systems AG.
“Depending on the process conditions, the SNCR unit is either installed directly in the combustion chamber or at the first section of the waste heat boiler. The question of the reducing agent is typically chosen depending on the plant size. Typically, smaller-sized units are operated with urea solution, larger-sized units are operated with ammonia water,” Walddörfer said.
Choosing between SCR and SNCR systems is almost entirely predicated on the emissions removal percentage required. “SCR and SNCR are drastically different in their capabilities,” said Ryan Hensel, senior proposal manager and subject matter expert for SCRs at Babcock & Wilcox (B&W). Hensel said SCR is commonly used in coal-fired, biomass, natural gas, and waste-to-energy applications. “We’ve shown 90-plus-percent removal on all of those applications. On an SNCR on a utility-scale boiler, you’re probably looking at somewhere more in the 30% to 40% range,” he said.
Yet, there are some applications in which SNCR can achieve higher removal percentages. “In some industrial applications, where they’re staying in the acceptable temperature range for longer, we’ve gotten removals up into the 60% to 70% range, but again, that’s the absolute right set of conditions,” Hensel said.
SCR Catalyst
Dan Johnson, vice president of Business Development with CORMETECH, explained that there are three elements that can be adjusted to create the optimal catalyst for any given SCR application. “One of them is the pitch,” he said. “Pitch is the size of the cell openings that allow gas to pass through the catalyst. If the gas has any sort of particulate in it, such as coal ash, you need to have the geometry of the catalyst such that it can pass that particulate.”
Johnson said it’s not uncommon to have cell openings of 7 millimeters (mm) on coal units to accommodate for ash concerns. CORMETECH also has catalysts made specifically for high-ash environments, where the cell openings are even larger but still maintain an extremely high catalytic surface area. In a gas-fired combined cycle unit, however, cell openings are generally much smaller—as low as 1.2 mm, 1.4 mm, or 2.1 mm. “And there’s everything in between, so it’s really important to pick the right pitch size for the particulate you might see in your flue gas,” he said.
The second item that must be considered is the catalyst formula itself. “On a coal unit, the formulas vary widely between units, because you’re trying to balance a couple of things. You’re adding catalytic metals to the catalyst to get the activity to reduce the NOx, but as you add more catalytic metals, it can oxidize more SO2 to SO3, so you’re designing the catalyst specifically for a particular unit to maximize activity of the catalyst with respect to NOx reduction, while minimizing activity on catalysts with respect to SO2 oxidation,” Johnson explained.
Gas units have set formulas that are commonly used. “Those formulas change, not with varying SO2 oxidation, but with varying temperature,” Johnson said. “As you go higher in temperature, you have to change the mixture of the catalytic metals that you’re using, because some of them will not perform well at higher temperatures. At CORMETECH, we pride ourselves on custom engineering our catalysts for every application, and it really is necessary to meet the performance requirements.”
The last consideration is the module design—the structure that surrounds the catalyst. “Every catalyst is packed or housed inside a steel frame. Then, that frame is installed into the unit, and there might be a lot of them—might be 140 of them in one layer in a coal unit, for instance,” said Johnson. Alterations to standard module designs are often based on gas flow variables (vertical vs. horizontal) and/or the desired pressure drop. The module design will change based on the performance target and to fit mechanically inside the unit.
Temperature Is an Important Variable
As previously mentioned, the operating temperature affects the performance of SCR systems. “The SCR usually operates within a temperature range of 300C to 350C [572F to 662F]. Depending on the offgas composition, lower temperatures can apply,” Walddörfer of Dürr Systems said. “If the exhaust air entering the system is not at the required temperature, it must be pre-heated using an air-to-air heat exchanger, for example. The air is then heated further by a burner or another type of heater until it reaches the catalyst operating temperature. In some specifications the SCR unit can directly be integrated in the waste heat recovery system—in between the boiler and economizer.”
“The SCR is kind of an optimization process on a lot of things,” said B&W’s Hensel. “The catalytic reaction is very temperature dependent. So, once you set a design temperature, if you go a little bit higher or a little bit lower, it will have some impact on the catalyst performance.”
In the case of some coal and gas-fired power plants, that can be a problem these days. As more wind and solar power has been added to the grid, operating requirements at many plants have changed. “As units have gotten away from baseloading and looked at more partial load or staying in partial load for longer, there are some consequences that come along with that. On the gas side, it’s a little bit easier, because natural gas not having sulfur in the gas, not having particulate in the gas stream, it really just becomes a little bit of variation in the performance of the catalyst itself,” Hensel explained.
“Maybe if you knew then what you know now, you would have picked a different temperature to optimize at, but typically in our experience, it’s a relatively minor impact on the removal percentage,” he said. “Where it can be more problematic is some application that has particulate or sulfur in the flue gas.
“On the particulate, if we design everything to keep all that particulate entrained—target specific fallout areas in the system where we can capture that—and then, for example, we’re running at half of that velocity, that might change some of those areas that we were specifically targeting before,” said Hensel.
“On the sulfur, at lower temperatures, ammonia can react with SO3 in the flue gas to form ammonium bisulfate. That’s a sticky substance that will plug up your catalyst and can have real performance issues on the catalyst in the short term. So, if a unit is running at 30% load and it was designed for 100%, you just need to be cognizant of staying at operating temperatures where you do you not see that phenomenon occur,” Hensel said.
Some possible retrofit options to mitigate problems associated with changing operating conditions include modifying catalyst cleaning equipment. “If the unit operating conditions lend themselves to more concentrated ash accumulation, typically along the front wall of the SCR reactor if the unit operates at lower loads for extended periods of time, we have gone into units and retrofit sootblowers or upgraded to something more powerful than we initially installed to target specific areas in the reactor,” Hensel noted.
“On the temperature side, we’ve done a number of solutions to increase the SCR operating temperature at lower load. Gas-side and water-side economizer bypasses are common. For example, B&W offers a V-Temp Economizer [Figure 1] that’s a patented water-side bypass. We have gone in and retrofit those on units to expand the acceptable operating range,” said Hensel.
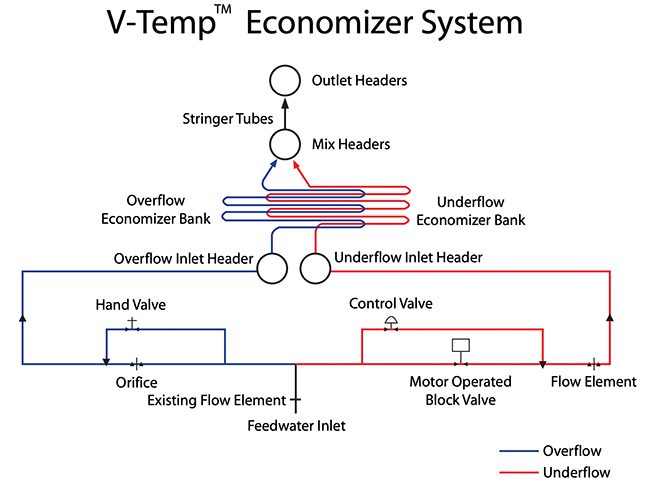 |
|
1. Babcock & Wilcox’s (B&W’s) V-Temp Economizer system is a unique design solution that allows boilers to operate at reduced loads with the selective catalytic reduction (SCR) system in service by maintaining the required minimum flue gas temperature at the SCR inlet. Courtesy: B&W |
Evolutionary Catalysts for Changing Needs
While SCR systems and most of the catalysts used in them have a long and successful operating history, there is still R&D work being done to improve performance. “CORMETECH has always prided itself on responding to issues in the market,” Johnson said. “When mercury became a problem for coal units, we developed a specific catalyst to help oxidize mercury so it can be captured. Ash became a problem as more people switched to different coals and now we have catalyst we call DUSTBUSTER that has very large openings and can pass a lot of ash, while maintaining very high catalytic potential.”
Johnson explained that on coal units the way the catalyst deactivates is through poisoning, not necessarily through loss of surface area. “Everyone in the SCR market uses a material called titanium dioxide for their base material. And that’s because that material, when it’s put in the right form, has a really good pore structure for catalytic reactions,” Johnson said. “What happens when you poison a catalyst is those pores get clogged or masked with poisons that latch onto the catalyst. And in a coal unit—we’re the only ones in the United States that do this—we can take back your coal catalyst, clean all those poisons off, reactivate it, and send it back out into service, and that creates a very low carbon footprint, it is economical, and that is unique to CORMETECH for the coal market. When that catalyst has reached the end of its regenerable life, we can then recycle the material avoiding any equipment going to the landfill.”
While CORMETECH has regenerated catalyst for customers during extended outages, the process is often better facilitated by having an extra layer in inventory. “We’ll have it in storage, regenerated, ready to go, and then when they have their outage, we’ll send them a layer. They send us the old one back, and then it’s ready for the next outage,” Johnson said.
On the gas side, CORMETECH claims its ELITE catalyst (Figure 2) offers the lowest pressure drop currently achievable in the industry. “No one can get lower than us,” Johnson said. “What that does is increase the turbine efficiency—you’re decreasing the backpressure on the turbine. And it also allows us to add a lot of activity without adding backpressure, which is hard to do without arranging the module in that sort of pleated fashion.”
 |
|
2. CORMETECH’s ELITE platform is an ultra-high surface SCR catalyst combined with ultra-high surface area module design, offering increased catalytic potential, decreased turbine backpressure, and reduced sensitivity to design changes. The combined technologies can accomplish pressure drop up to 75% less than traditional designs with the same or better performance. Courtesy: CORMETECH |
Meanwhile, the company also has special formulas for other unique scenarios. “If you have high NO2, we have a catalyst that’s specifically designed for that, because NO2 is harder to reduce than NO,” said Johnson. “If you want a catalyst that can do CO oxidation and NOx removal in one layer, we have that. It’s called our METEOR or multi-emission catalyst,” he said. “Designs get very specific for these units and we try to create a partnership with the utility or operator to maximize performance.”
—Aaron Larson is POWER’s executive editor.