Browns Ferry Unit 1 Restart: World-Class ALARA Performance
TVA completed the $1.9 billion restart of the 1,100-MW Browns Ferry Unit 1 in 2007. That restart project provided the opportunity to incorporate state-of-the-art materials and radiation-reduction techniques to ensure that Unit 1 would be an industry-leading low-ALARA-exposure plant when it returned to service. The reductions achieved were significant.
Tennessee Valley Authority (TVA) became a nuclear utility when the Browns Ferry Nuclear Plant (BFNP) Unit 1 entered service on August 1, 1974. By March 1977, the plant had grown to three 1,000-MWe units, making it the largest nuclear plant in the world at the time. The plant, named after a ferry that once operated nearby, is located on an 840-acre site beside Wheeler Reservoir on the Tennessee River near Athens, Alabama (Figure 1).
 |
| 1. Full service. With the 2007 restart of Unit 1, Tennessee Valley Authority’s three-unit Browns Ferry Nuclear Plant is now back in full service. Source: TVA |
Following the infamous 1985 plant fire, TVA shut down all three units, indefinitely. After completing necessary repairs and plant safety upgrades, Units 2 and 3 reentered service during 1991 and 1995, respectively. Unit 1 remained in long-term shutdown until a $1.9 billion restart project was approved by TVA’s Board of Directors in 2002. Unit 1 reentered service in 2007. That same year, the American Nuclear Society recognized the unit with its Utility Achievement Award for Outstanding Improvement in Performance, “in recognition of the most extensive restart effort in the nuclear industry.”
BFNP features three General Electric boiling water reactors (BWR). After restart, the BWR/4 reactors produced about 1,100 MWe, after completing Nuclear Regulatory Commission (NRC)–approved “stretch” uprates on each unit. Today, BFNP has applications pending for a 14.3% thermal uprate for each of the three units. For more on the history of BWR design evolution, see “The Evolution of the ESBWR” in POWER’ s online archives at https://www.powermag.com.
As part of the Unit 1 restart project, state-of-the-art materials and techniques were to be incorporated to ensure that it would be an ALARA (as low as reasonably achievable) radiation exposure plant when it returned to service. The remainder of this article discusses the program approach and specific improvements made to components and systems to achieve an industry-leading low ALARA exposure rate.
Early Program Planning
The minimum requirement for ALARA exposure for the Unit 1 restart project was to match the radiation levels of the other two BFNP units. The first step was to form a Cobalt Reduction High Intensity Team (HIT) with representatives from the Radiation Protection, Chemistry, Engineering, Maintenance, Modifications, Planning, and Procurement groups. The next order of business was an evaluation of each group’s radioactive cobalt (Co-60), reduction plans.
Clearly, one of the best methods for reducing the source term for radioactive cobalt is to reduce cobalt-bearing materials. The second-best approach is to control and mitigate the activity from plant out-of-core surfaces. Browns Ferry utilized both approaches for the Unit 1 restart program.
During August 2003, TVA asked the Electric Power Research Institute (EPRI) to perform an independent assessment of the BFNP Unit 1 restart plan. The goal of this evaluation was to determine measures that would assist TVA in making the restart world-class for cobalt dose rate control. The goal was to use all of the tools, technologies, and guidance that EPRI had compiled to date to reduce Unit 1’s radiation source term for radioactive cobalt.
EPRI’s initial assessment identified several areas that required further investigation, but unfortunately, much of the design work for many plant systems had already been completed and approved by the NRC. At that point, any new design changes had the potential to delay the projected plant restart date of May 2007. However, adequate time and resources were available for TVA to implement a number of the EPRI suggestions.
One important EPRI recommendation was that, at a minimum, all primary system piping should be electropolished (EP) and pre-oxidized to minimize radionuclide deposition on piping surfaces. For forged piping, the interior surface is smooth, and electropolishing alone provides a limited reduction in true surface area. Thus, it is imperative that pre-oxidation follow electropolishing. If the schedule permitted, it was recommended that the reactor recirculation pump bowls and large system valves be electropolished and pre-oxidized. In this latter case, if pre-oxidation was not possible, at least the electropolishing should be done, as some benefit is realized for these cast stainless steel surfaces. Unlike forged piping, the cast surfaces of pumps and valves are very rough, and electropolishing provides a reduction in the true surface area.
The best surface pretreatment for new components is the EPRI stabilized chromium process (SCrP). However, that recommendation encompassed a large percentage of the plant primary system components and was not possible considering the tight project schedule. Instead, SCrP was used on carefully selected valves, piping, pump casings, impellers, valve bodies, and process radiation monitor housings (see EPRI 1003373, Stabilized Chromium Process Final Report, August 2002).
After performing a chemical decontamination to reduce worker exposure, the majority of the major piping systems in Unit 1 were removed for replacement, and at one point it was even possible to walk into the drywell in street clothes.
Source Term Elimination
The EPRI SCrP consists of three discrete steps.
First, the component is electropolished. Electropolishing wetted surfaces reduces the true surface area of the component by creating a micro-smooth finish, as observed at a magnification of greater than 100x. Mechanical polishing of metal surfaces does improve the finish, but it leaves rough edges and asperities. These rough edges or pinnacles then have a greater corrosion potential than the base materials would normally exhibit. Electropolishing preferentially removes the pinnacles and asperities while creating a microscopically smooth surface. Also, fewer activated corrosion products are deposited on the smooth surface layer than would be found in the micro-crevices created from mechanical polishing alone.
The second step is chromium application. To ensure that the surface is clean for the chromium application, it immediately follows the electropolishing step. A thin, 3,000- to 13,000-angstrom layer of chromium is applied to the wetted surface. This layer of chromium provides a chromium-rich corrosion inhibition layer.
The final step is passivation or pre-oxidation. Pre-oxidation of electropolished and/or chromium-enriched surfaces allows for the initial corrosion film to be formed in a controlled environment. The corrosion film at the component surface will be free of radionuclides. With the reduced corrosion potential, subsequent activated corrosion products will build up at a much lower rate.
Stabilized chromium surfaces are most passive in reducing chemistry environments, which are typical of pressurized water reactor primary systems and BWR under hydrogen water chemistry (HWC) and noble metals application.
The following BFNP Unit 1 components had SCrP applied prior to reinstallation.
RWCU Pumps. The SCrP-treated reactor water cleanup (RWCU) pumps that were intended for Unit 1 service were installed in Units 2 and 3 when those pumps needed replacement, as there was time in the schedule to acquire new pumps for Unit 1. While installed in Units 2 and 3, the dose rate rise was very low, and, interestingly, higher flow rates at normal operating amperages were observed. Increased RWCU flow is desirable for BWR plants because it has the potential to reduce steady state operating impurity levels in the reactor coolant. It is believed that the smoother surfaces and minimization of high and low spots in the flow path result in lower pressure drop across the pump and, therefore, greater flow.
Figure 2 illustrates the SCrP application on a pump impeller and casing. Photographs of the SCrP application steps on a RWCU valve are shown in Figure 3.
 |
| 2. RWCU pump SCrP application. Courtesy: Radiological Solutions Inc. |
 |
| 3. RWCU valve internals. Shown are the as-received (top), electropolished (center), and post-application states of the SCrP (bottom). Courtesy: Radiological Solutions Inc. |
RHR Testable Check Valve. The residual heat removal (RHR) system testable check valves were identified by the ALARA staff to be high dose contributors on Units 2 and 3, as these valves historically have required frequent maintenance. SCrP was applied to the valve internals (Figure 4) and to the inside and outside of the valve bodies (Figures 5 and 6).
 |
| 4. SCrP applied to valve internals. Courtesy: Radiological Solutions Inc. |
 |
| 5. SCrP applied to the inside of a valve body. Courtesy: Radiological Solutions Inc. |
 |
| 6. SCrP applied to the outside of a valve body. Courtesy: Radiological Solutions Inc. |
Reactor Recirculation System Piping. The reactor recirculation piping was electropolished and pre-oxidized. A section of large-bore piping is shown in Figure 7.
 |
| 7. Large-bore piping electropolished and pre-oxidized. Courtesy: Radiological Solutions Inc. |
RWCU Piping. All of the RWCU piping was electropolished and pre-oxidized.
Non-regenerative Reactor Water Cleanup Heat Exchanger. RWCU heat exchangers historically are also high-dose-rate components. The non-regenerative RWCU heat exchanger was electropolished and pre-oxidized during manufacturing (Figure 8).
 |
| 8. RWCU non-regenerative heat exchanger electropolished and pre-oxidized. Courtesy: Radiological Solutions Inc. |
Other System Improvements
The HIT also pursued other reductions of cobalt sources in the plant, including those that follow.
Replaced Stellite Surfaces. A total of 78 valves were replaced with non-Stellite trim or non-cobalt-bearing hardfacing. The HIT team based valve replacement decisions on severity of service (steam or water, location, flow to the reactor pressure vessel, and normal operating position) and size. Valves such as check valves that slam open and closed on their seats, as well as valves that operate in a throttled position, were likely candidates for replacement.
All 185 control rod blades were replaced with non-Stellite materials (pins/rollers). Many plants are replacing old control rods with Stellite-free components as their normal service cycle permits replacement. This in-core source of cobalt is claimed to account for up to 20% of the cobalt source term in many plants.
In addition, the low-pressure turbine buckets were installed with flame-hardened buckets in lieu of Stellite.
Reduced Recontamination Rates. Corrosion rates on new components, including piping, are the highest when the plant initially starts up or returns to service. A protective corrosion layer is soon formed on the material surface, and then the corrosion rate slows considerably. Any impurities that are present in the coolant as the new corrosion layer is forming have the potential to become incorporated into the oxide layer. If the impurities are radioactive, then the activated corrosion products contribute to the component’s radiation dose field. The ideal situation is to form a protective corrosion layer with no radioactive corrosion products (that is, pre-oxidize).
The U.S. BWR fleet has experience with high recontamination rates. The recontamination rate for fresh metal surfaces may be much greater than for previously installed or original piping (Figure 9). Forming a protective corrosion layer on component surfaces prior to installation significantly minimizes the amount of activated corrosion products that are initially incorporated into the new surface corrosion films.
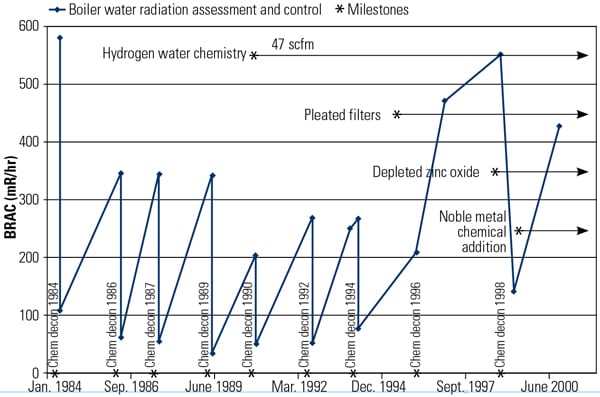 |
| 9. Typical BWR reactor recirculation piping recontamination response following chemical decontamination. Source: Radiological Solutions Inc. |
Changed Condenser Tubing. The condenser tubing was changed from admiralty brass to 304 stainless steel. This change will assist with minimizing/eliminating reactor coolant–soluble copper, which is a known contributor to localized fuel failures. High reactor coolant copper also can also interfere with reaching electrochemical protection under HWC conditions. However, “low cobalt” content of the tubing was not specified because the assessment and replacement process began after the materials were purchased and on site. Browns Ferry has a filter demineralizer condensate polishing system that is less efficient for removing soluble contaminants than deep bed counterpart plants. In any case, retubing the condenser with a lower cobalt–containing metal was no longer feasible.
Cleaned 92 Reload Fuel Bundles. Although new fuel would have been preferable, as a cost-mitigating action, 92 reload assemblies (36 once-burned and 56 twice-burned) from Unit 2 were cleaned using EPRI ultrasonic fuel cleaning technology and loaded into the core for plant restart. The cleaning removed an estimated 4,485 curies of cobalt that would have been inventory for redistribution to other plant surfaces following plant restart. Figure 10 shows photos of a fuel assembly before and after the corrosion products were removed from the reload fuel.
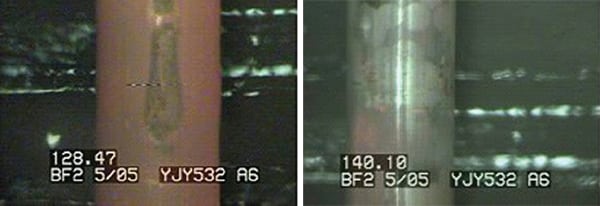 |
| 10. Reload fuel prior to (left) and after (right) ultrasonic cleaning. Courtesy: Radiological Solutions Inc. |
Upgraded Condensate Filtration. Unit 1 returned to service using pleated filter elements in all of the condensate filter demineralizer vessels. This assisted in controlling feedwater iron down to the recommended range of 0.1 to 1.0 ppb under HWC/noble metal chemical addition (NMCA) conditions, per EPRI 2008 BWR Water Chemistry Guidelines.
Changed Depleted Zinc Oxide (DZO) Start. DZO is added to the feedwater at the majority of BWR plants for dose rate control. Keeping the reactor coolant–soluble Co-60/soluble zinc ratio <2.0 x 10-05 µCi/ml/ppb is the target level, per EPRI guidelines. The amount of zinc added to the feedwater is typically limited to 0.4 ppb by the fuel vendors. Zinc addition was not started at Unit 1 until three months after startup. This delay was required for fuel preconditioning. The best condition for dose rate mitigation would have been to have an inventory of soluble zinc available during initial startup to compete with cobalt for incorporation into the corrosion film(s).
Reduce Soluble Cobalt. The soluble fraction of activity in the reactor coolant is defined as the portion that will pass through a 0.45-µm filter. Plants with low soluble reactor coolant Co-60 tend to have the lowest piping dose rates. Unit 1 implemented HWC and noble metals controls during the first fuel cycle, in conjunction with the other condensate mitigation strategies described above. The results of this comprehensive treatment program, shown in Figure 11, are encouraging. Most plants that have implemented these mitigation strategies with a higher source term have experienced increased piping and out-of-core dose rates following initial startup, unlike Unit 1.
 |
| 11. Browns Ferry Unit 1 reactor coolant–soluble cobalt trend. Source: Radiological Solutions Inc. |
Other Best Practices. Many other innovative tools and practices were developed by BFNP staff and were incorporated into the Unit 1 recovery plan. These tools and practices have subsequently been shared with the industry, effectively aiding exposure reduction programs at other sites. For example, x-ray fluorescence measurements of final cleaning wipes from equipment before closure ensure that cleaning has removed all of the residual elemental cobalt.
Excellent ALARA Program Results
One metric that determines the success of a program focused on radiation reduction is measured contact dose rates. The Boiling Water Radiation Assessment and Control (BRAC) point surveys resulted in an average contact dose rate of 15 mR/hr. These readings are with the piping filled and insulation installed. For comparison, after one cycle following chemical decontamination, Unit 2 average BRAC point dose rates were 200 mR/hr. General area dose rates for Unit 1 were approximately 3 to 5 mR/hr, and for Unit 2 were 10 to 20 mR/hr. This low rate of activity is a significant improvement over the buildup typical of plants that have not implemented source term reduction efforts (Figure 9).
Although it is difficult to normalize outage performance data because of the large number of variables, another measure of dose performance is the effective dose rate (EDR), which is the accumulated dose divided by the number of hours worked in the drywell (Table 1). The total exposure for the first refueling outage was 45 rem, corrected to remove extended power uprate (EPU) work on the steam dryer.
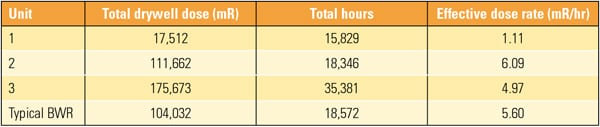 |
| Table 1. BFNP Unit 1 outage effective dose rate. Source: Radiological Solutions Inc. |
Other Lessons Learned
In addition to the previously mentioned upgrades, the following list of other source reductions should be helpful for other plants considering setting up an ALARA program:
- Start DZO addition prior to startup. An inventory of soluble zinc in the reactor coolant is required to compete with cobalt for sites when forming the corrosion layer.
- A lower cobalt specification for condenser tubing material would have reduced the cobalt source term for the plant. High performance levels in the condensate filter in the demineralizer system can minimize the effect from condenser tubing general corrosion.
- Jet pump hold-down beams/wedges have been identified as an in-core source of cobalt at some plants. Unfortunately, Unit 1 was not able to change the wedges to non-cobalt-bearing material.
- Iron injection may have been useful to assist with “sticking” corrosion products to the fuel surfaces. Some iron is required for this purpose, and other plants with extremely low feedwater iron intentionally inject iron into the feedwater to control reactor coolant–soluble cobalt.
- Reactor recirculation pump maintenance is labor-intensive, and workers may be exposure-limited. On Unit 1, there was no time to mitigate this exposure because of the tight restart schedule.
- Take the time to develop methods to perform EP/pre-oxidation on components in situ. This will allow for more components to have ALARA techniques implemented and can further reduce occupational exposure. â–
— Richard Kohlmann (rkohlmann@ radiologicalsolutions.com) is president and Roger Asay is principal consultant for Radiological Solutions Inc. Steve Cowan is a health physicist, Mike Scarboro is ALARA supervisor, and John Underwood is the chemistry manager at TVA’s Browns Ferry Nuclear Plant.