In Part I of this three-part series, "Improving the Performance of Boiler Auxiliaries," we explored the air preheater (APH) and important performance calculations. Part II examines performance degradation resulting from corrosion and fouling that is caused by coal combustion flue gas constituents plus the effects of ammonia and sodium bisulfite injection for SO3 mitigation.
Formation and Deposition of Sulfuric Acid
Sulfuric acid (H2SO4) is formed as a vapor in the boiler by the association of sulfur trioxide (SO3) with water vapor. In the 600F to 660F range, both SO3 and H2SO4 are present in the flue gas, but H2SO4 is dominant. Below 600F, SO3 is almost completely converted to H2SO4.
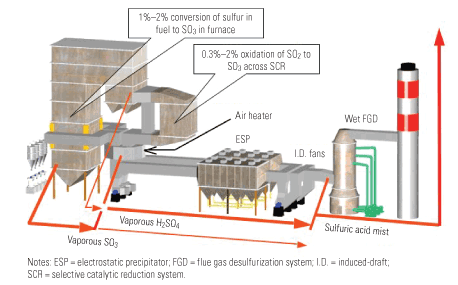
The SO3 is initially formed by the reaction of sulfur dioxide (SO2) with atomic oxygen (O) in the post-flame region of the furnace and by the catalytic (heterogeneous) oxidation of SO2 with molecular oxygen in the convective pass of the boiler. The former is a homogenous reaction, where the amount of formed SO3 depends on the excess O2 level. With lower excess O2, less SO3 is formed in the furnace. For the excess SO3 level, measured at the economizer, in the 2% to 6% range, the SO3 vs. O2 relationship is almost linear.
Typically, 0.7% to 1% of SO2 is converted to SO3 through homogenous reactions. Heterogeneous reactions are responsible for approximately 1% conversion of SO2 to SO3, but the actual conversion ratio, as discussed below, depends on many factors. Some of the SO2 (0.3% to 2%) is oxidized to SO3 within passages of the selective catalytic reactor (SCR, see Figure 1). The increase in the amount of SO3 leaving the SCR is a function of the oxidation level of its catalyst: Very low oxidation catalysts raise the SO3 level only slightly, but high-oxidation catalysts can double inlet SO3 concentration. Application of advanced SCR catalysts will reduce the SO2 to SO3 conversion in the SCR to 0.1%.

1. Oxidation of SO2 to SO3 in a boiler and SCR. Source: Electric Power Research Institute
Besides the excess O2 level, there are other design and operating parameters that affect SO3 formation. Among these are the fuel sulfur content, coal mineral content and composition, convective pass surface area, flue gas and tube temperatures, and coal fineness. Thus, the same fuel burned in two different units, or in the same unit under two different operating conditions, can produce substantially different levels of SO3.
As the flue gas is rapidly cooled by the sprays of liquid in the flue gas desulfurization (FGD) system, the vaporous sulfuric acid undergoes a shock condensation process that produces very fine sulfuric acid aerosol particles. These aerosol particles, for the most part, are too small to be effectively captured in the FGD system and are emitted into the atmosphere as a sulfuric acid mist. The optical light-scattering effects of sulfuric acid aerosol particles produce a visual discoloration of the plume exiting the power plant stack. The minimum SO3 concentration necessary for causing this phenomenon depends both on atmospheric conditions (the direction of sunlight, temperature, humidity, and wind speed) and stack specifics (stack diameter and exit gas velocity). Flue gas SO3 concentrations of about 10 ppmv can result in plume opacities greater than 50% in some cases; at 5 ppmv, the opacity is about 20%. The specific SO3 concentration at which a blue plume can be seen is a function of atmospheric conditions and stack characteristics. However, it is generally accepted that if the SO3 concentration is less than 5 ppmv, there are no visible discoloration effects. It has to be noted that the term "SO3" used here refers to varying proportions of vaporous (gaseous) SO3 and sulfuric acid and to sulfuric acid aerosol particles downstream of the wet FGD system.
Not all of the formed SO3 is discharged through the stack into the atmosphere. Some SO3 or vaporous sulfuric acid will condense on fly ash particles and ends up in the APH or electrostatic precipitator (ESP). The quantity, surface area, and alkali content of the ash, as well as the temperature of the flue gas, determine how much SO3 is removed (from 20% to considerably higher than 50%).
Regenerative APHs add another variable because of variations in the exit flue gas temperature. Depending on the inlet SO3 concentration, the average gas temperature, rotational speed of the APH matrix, and the thickness of the heating element, the temperature of the flue gas in the cold end layer of the APH can be significantly below the acid dewpoint. As a result, condensation of sulfuric acid occurs on the APH heat transfer surfaces.
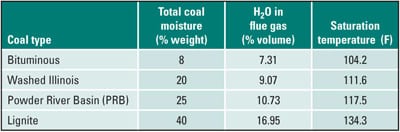
As the flue gas and metal temperature decrease below the acid dewpoint, condensation of sulfuric acid begins to occur. An example is provided to illustrate the location and size of the acid deposition zone. The axial variation of maximum and minimum metal temperatures is presented in Figure 2. At an axial location of approximately 17 inches from the APH cold end, the minimum metal temperature decreases below the acid dewpoint temperature (268.2F in this example), and sulfuric acid begins to condense on the APH heat transfer surfaces in the hot end (HE) and cold end (CE) layers of the APH. This dewpoint temperature corresponds to SO3 concentration in the flue gas of 5 ppmv and flue gas moisture content of 13.7% volume (Figure 3).
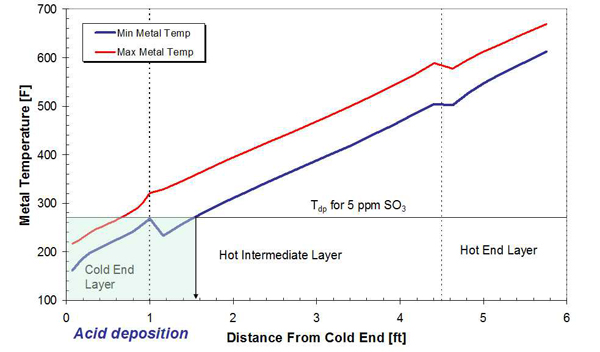
2. Acid deposition zone in a Ljungstrom APH with new baskets and operating at design conditions. Source: Energy Research Center (ERC)
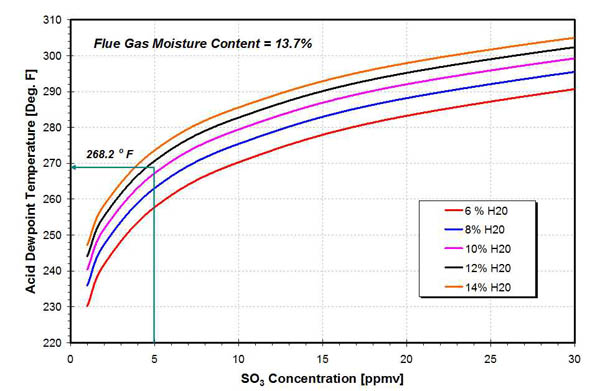
3. Acid dewpoint as a function of SO3 and H2O concentration in the flue gas. Source: ERC
As the APH matrix rotates through the flue gas duct (gas cycle), the matrix temperature increases as heat is transferred from the hot flue gas. In the region of the APH close to the cold end, metal temperature will increase above the acid dewpoint temperature, as presented in Figure 4. As the matrix is rotating through the air duct (air cycle), the metal temperature decreases as heat is transferred to the cold combustion air.
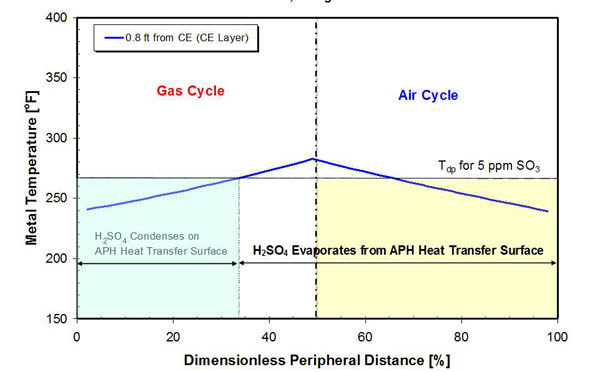
4. Acid deposition and evaporation zones in an APH with new baskets and operating at full load at design conditions. Source: ERC
Acid condensation will occur in the region where the metal temperature is below the acid dewpoint temperature (approximately 35% of the rotation cycle). As the metal temperature increases above the acid dewpoint temperature, deposited sulfuric acid will start to evaporate from the metal matrix. Evaporation will continue as the APH matrix is rotating through the air duct due to a difference in partial pressure of sulfuric acid at the surface and in the combustion air stream. The extent to which this evaporation process occurs is primarily a function of the moisture content of the combustion air (with higher moisture reducing evaporation) and the matrix temperature. The actual occurrence of acid evaporation has been verified in field measurements.
The acid deposition and evaporation zones during the gas cycle are shown in the APH deposition map, Figure 5. The results show that, although acid deposition starts in the HE layer of the APH, the area affected by deposition is relatively small. In comparison, a very large deposition zone is set up in the CE layer of the APH. The "no acid deposition" zone in the CE layer denotes the region of the APH where, as a consequence of matrix rotation, metal temperatures increase above the dewpoint.
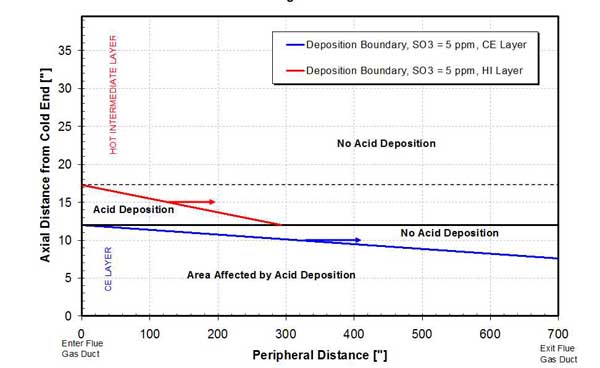
5. APH acid deposition map when operating at design conditions. Source: ERC
It has to be noted that in the area of the APH operating below the acid dewpoint, maximum corrosion occurs in the zone operating approximately 30F below the acid dewpoint temperature (Figure 6). The reason for a higher corrosion rate at lower temperatures is that the acid deposition rate is highest in that area; the rate of condensation of sulfuric acid on the APH matrix depends on its concentration and temperature depression (the difference between the metal and acid dewpoint temperatures). As sulfuric acid condenses out of the flue gas stream, its concentration in the flue gas decreases, resulting in maximum acid deposition rate.
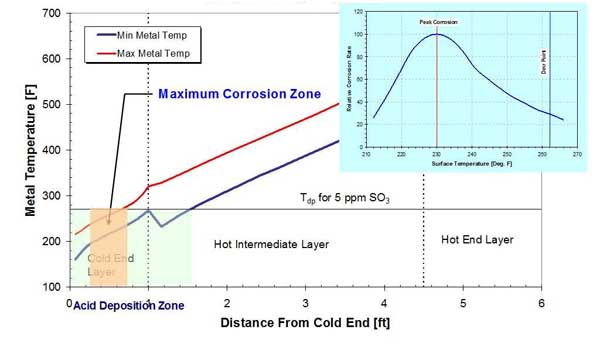
6. Maximum corrosion zone in the APH with new baskets and operating at design conditions. Source: ERC
Formation and Deposition of Ammonium Bisulfate
Experience in the U.S. and elsewhere has shown that conventional regenerative APHs are vulnerable to fouling with ammonium bisulfate (ABS) deposits, resulting from the operation of ammine-based NOx-removal equipment, such as SCR and selective noncatalytic reduction (SNCR) reactors. Though it was not thought to be a problem, assessments of this subject have uncovered that the extent of this APH fouling problem in the U.S. is wider and more serious than expected. This is in part due to the burning of high-sulfur, low-alkali coals; high values of SCR NOx removal efficiency; and inherent ammonia slip problems associated with SNCR reactors under cycling conditions.
Revised APH fouling criteria are now specifying that SO3 levels lower than 2 ppmv to 3 ppmv and ammonia (NH3) levels lower than 1 ppmv to 2 ppmv are required to avoid APH fouling by ammonium salts. Unfortunately, both NH3 and SO3 are present in the flue gas downstream of the SCR or SNCR reactor. NH3 slip is practically an unavoidable consequence of injecting ammonia or urea into the flue gas for NOx reduction. As described in the previous section, SO3 is formed from SO2 by the homogeneous oxidation reaction in the furnace and heterogeneous oxidation reactions in the convective pass of the boiler and within the SCR catalyst passages. The SCR catalyst is also responsible for increased SO2 to SO3 conversion, which further aggravates the fouling problem.
The SCR process involves the injection of ammonia into the flue gas, where it reacts with NOx in the presence of a catalyst to form molecular nitrogen (N2) and water. In most cases, the SCR is located upstream of the APH and upstream of the particulate control device and operates in a high-dust environment. As the ammonia injection rate increases, the probability of the denitrification reaction increases and, consequently, SCR performance improves. However, excessive reagent injection may result in ammonia slip (Figure 7).
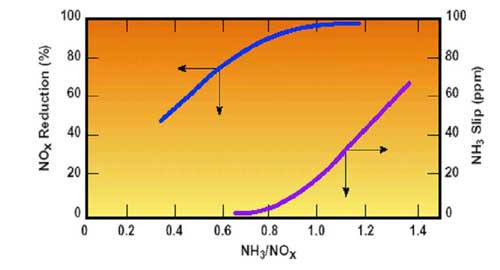
7. Effect of NH3/NOx molar ratio on NOx reduction and total ammonia slip (NH3 on fly ash and in flue gas) in an APH. Source: ERC
Deterioration of SCR performance due to catalyst poisoning (Figure 8) may also result in ammonia slip. Therefore, after a period of time in an SCR reactor that was tuned for zero ammonia slip when the catalyst was new, ammonia slip will occur as catalyst activity is reduced.
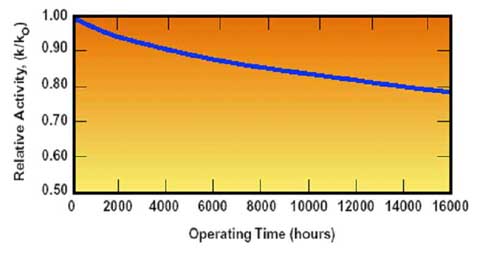
8. Decrease in catalyst activity due to aging. Source: ERC
SNCR technology also involves injection of ammonia or urea into the flue gas to react with NOx, but in this case, it’s without the use of a catalyst. For the reaction kinetics to proceed, the ammonia must be injected into the flue gas at a higher temperature than with an SCR. Typically, the ammonia is injected in the upper furnace region of the boiler, where the gas temperature is in the range of 1,800F to 2,000F.
Flue gas temperature has a large effect on SNCR performance and ammonia slip. For the maximum denitrification efficiency, the reagent (typically urea) has to be injected within a relatively narrow temperature window (Figure 9). However, a relatively small decrease in flue gas temperature from optimal value will result in a sharp decrease in NOx reduction and a large increase in ammonia slip. The higher the reagent injection rate, the better the SNCR performance and the higher the ammonia slip.
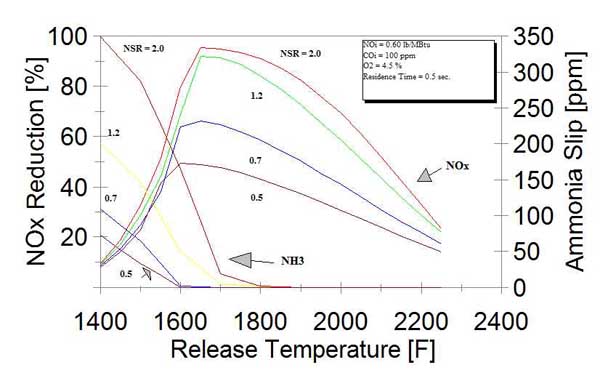
9. Effect of temperature on SNCR performance and NH3 slip. Source: ERC (Carlos Romero)
In SNCR systems, the unreacted NH3 is largely a result of operation at non-optimal conditions. Such conditions might include, for example, an untuned reagent injection system, heavy unit load duty cycle, changes in boiler operating conditions that result in changes in flue gas temperature at the location where reagent is injected, or boiler operational upsets.
When ammonia slip occurs, ammonium sulfates form in the presence of SO3 in the flue gas. Once formed in the flue gas, ammonium compounds deposit on the APH heat transfer surfaces (typically in the intermediate layer of the APH) and increase flow resistance. This results in increased APH pressure drop and fan power requirements and a reduction in net unit power output. An excessive increase in APH pressure drop could result in unit derates due to fan power limitations and cause unscheduled unit outages for APH cleaning.
The reaction of NH3 and SO3 in the presence of water vapor begins at elevated temperatures (550F to 650F). The principal reactions are:
NH3 + SO3 + H2O • NH4HSO4 (ammonium bisulfate, ABS) [Equation 1]
NH3 + H2SO4 • NH4HSO4 (ABS) [Equation 2]
NH3 + NH4HSO4 • (NH4)2SO4 (ammonium sulfate, AS) [Equation 3]
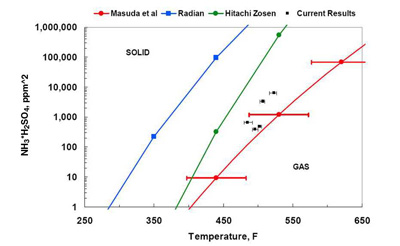
Formation of ABS and AS depends on the NH3 to SO3 molar ratio. If flue gas contains more NH3 than SO3 (on a molar basis), under equilibrium conditions, formation of AS is favored. If the SO3 concentration exceeds the NH3 concentration, ABS formation is favored, again under equilibrium conditions. However, kinetics also plays a role: The ABS formation rate is higher compared to the AS formation rate. Therefore, in many cases, ABS is a predominant product. ABS is hygroscopic, corrosive, sticky, and difficult to remove in its solid state. Its formation temperature depends on the inlet SO3 and NH3 concentration. The ABS formation temperature, predicted by correlations developed by Hitachi-Zosen and Radian/EPA, are presented in Figure 10.
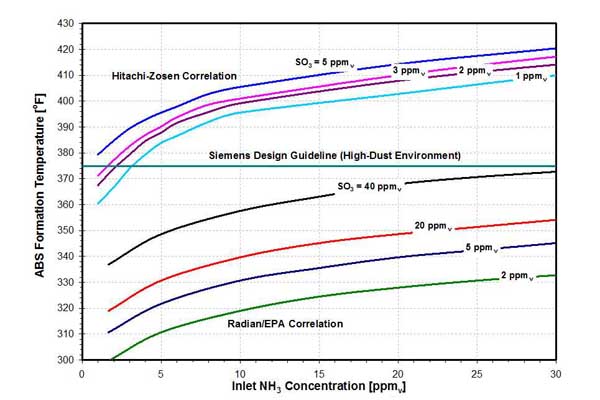
10. ABS formation temperature as a function of inlet NH3 and SO3 concentration. Source: ERC
Although both correlations predict increase in formation temperature as inlet concentrations of the reagents increase, there is a significant discrepancy between calculated ABS formation temperatures, with Hitachi-Zosen predictions being substantially (approximately 75F) higher. One of the reasons for this difference involves reactions between NH3 and SO3. It seems the Hitachi-Zosen correlation was developed by considering the reaction of NH3 and SO3 [1] while the Radian correlation was developed by considering the reaction of NH3 and H2SO4 [2].
At typical flue gas temperature levels upstream of the APH, the vapor-phase H2SO4 is a predominant species (below 600F); SO3 is almost completely converted to H2SO4, as discussed in the previous section. This suggests that from an equilibrium point of view, the more representative reaction is that of NH3 and H2SO4, and the Radian/EPA correlation should provide better predictions of ABS formation temperature than Hitachi-Zosen. However, this issue is not completely settled, and additional studies performed by other researchers have introduced additional uncertainties (Figure 11). Due to these uncertainties, some manufacturers use a fixed value of ABS formation temperature. For example, Siemens uses 375F as a design guideline.
11. Comparison of ABS formation temperature data. Source: EPRI
Ash loading also plays a significant role in the ABS formation and deposition processes. This is because a portion of the NH3 and SO3 present in the flue gas is adsorbed on the fly ash particles. This lowers their concentrations and reduces the formation temperature. Also, at lower concentrations, the amount of ABS and AS formed is less. The amount of fly ash is much larger, compared to the amount of ABS and AS formed, typically 20:1. Therefore, there is plenty of ash to mechanically remove (scrub) a portion of the deposited ABS layer from the APH heat transfer channel walls. Field experience shows that APH fouling under low-dust conditions (that is, with a hot-end ESP) is higher by a factor of three to four compared with high-dust situations (a cold-end ESP). Although it has not been substantiated by laboratory- or pilot-scale experiments, ABS formation temperatures predicted by the Radian/EPA correlation are in better agreement with field observations, most likely due to the fly ash effects and more-representative reaction. The ABS formation temperature predicted by the Radian/EPA correlation is for convenience also presented as a function of the inlet SO3 and NH3 concentrations in Figure 12.
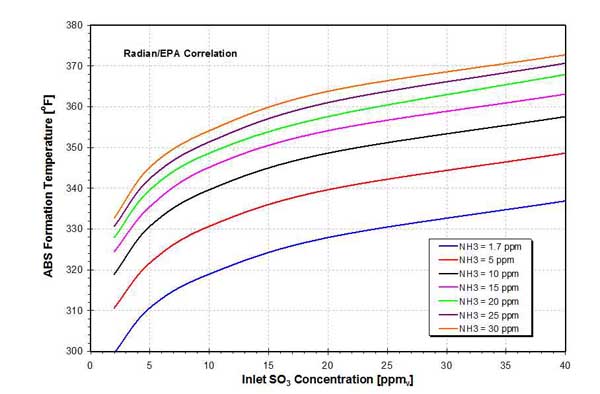
12. ABS formation temperature as a function of inlet SO3 and NH3 concentration (Radian/EPA correlation). Source: ERC
Precipitation of ABS from the gas state will continue until its freezing point is reached (approximately 300F), establishing a narrow formation temperature window typical of the intermediate APH layer baskets. The ABS deposition zones, determined by using Siemens design guideline and Radian/EPA correlations to predict formation temperature, are presented in Figure 13. The results show that the ABS deposition zone is very narrow, especially when using inlet concentrations of SO3 and NH3 to predict formation temperature. Inlet SO3 and NH3 concentrations of 5 ppmv were used in this example.
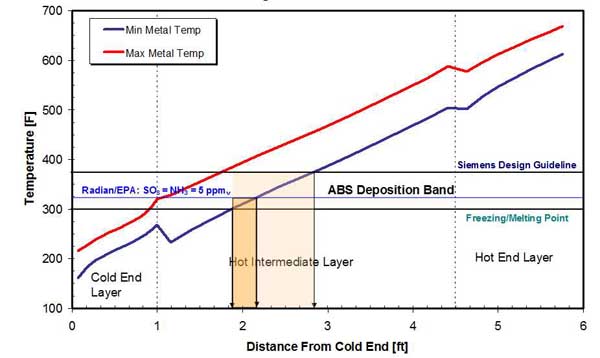
13. ABS deposition zone in a typical APH in a high-dust installation. Source: ERC
The problem of ABS deposition on APH heat transfer passages is further complicated by the fly ash capture on the sticky ABS layer and the deposition of sulfuric acid (H2SO4), as shown in Figure 14.
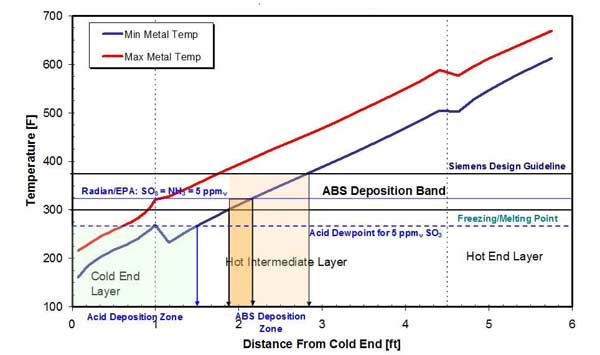
14. Acid and ABS deposition zones in a typical APH in a high-dust installation. Source: ERC
APH design characteristics can also aggravate the ABS fouling problem. These are related to the characteristics of the heat transfer surfaces (surface material and finish) and characteristics of the APH elements (axial layer splits, depth of the CE layer, and design of the heat transfer elements/channels). The presence of the axial layer split in the ABS deposition zone results in ABS being deposited on the upstream face of the layer. This results in very high rates of APH pressure drop buildup. Also, the layer split reduces sootblower effectiveness. Field experience in Europe, Japan, and the U.S. shows that a deep CE layer, containing both ABS and acid deposition zone in one layer, could be very effective in controlling APH fouling. However, basket replacement represents a costly solution to the APH fouling problem.
Other contributing factors are characteristics of the APH sootblowing equipment and their activation schedule, such as the operating profile of the unit (for example, base load or load cycling) and the SCR or SNCR system (for example, maximum NOx reduction and seasonal differences).
Ammonium sulfate is formed as a dry, non-sticky powder. AS particles are small (less than 10 microns), difficult to remove in the ESP, and could contribute to PM10 emissions and opacity.
APH Fouling Caused by Combustion of Low-Rank Coals
Low-rank coals, such as Western coals and lignites, differ from eastern and other bituminous coals in many aspects, including: higher reactivity, lower nitrogen and sulfur content, high refractory properties, and inherently high calcium (and other alkali) content. A comparison of ash mineral content for lignite and bituminous coals is presented in Figure 15.
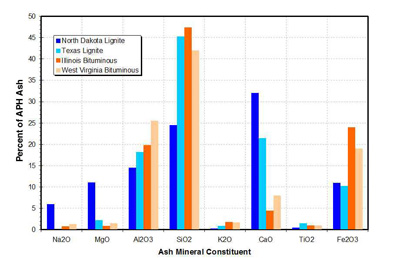
15. Ash composition by coal rank. Source: ERC
The mineral contents of lignite and Powder River Basin (PRB) coals are compared in Figure 16. These differences in mineral content cause significant differences in slagging and fouling properties between the bituminous coals and PRB coals and lignites.
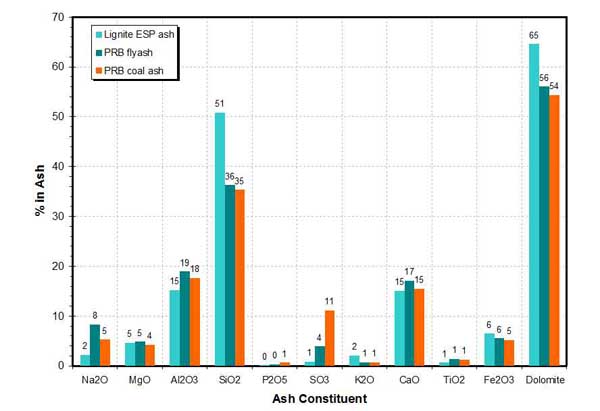
16. Ash composition of lignite and PRB coals. Source: ERC
For example, the high refractory properties of low-rank coals result in higher-temperature flue gas at the APH inlet and outlet, which pushes acid and ABS deposition zones closer to the cold end of the APH. Inherently low sulfur content results in lower SO2 and SO3 concentrations in flue gas. However, higher coal moisture content results in higher moisture content in flue gas.
Also, low-rank coals have high calcium (Ca) or lime (CaO) content, in excess of 20% (Figure 15). Ca is present in different forms; however, the bulk of Ca is in a fine reactive form, which is oxidized during combustion and released as very fine CaO particulates or fume. CaO reacts with SO2 and SO3 from flue gas, forming calcium sulfate (CaSO4). Also, dolomite accumulates as "popcorn" ash at the APH and SCR inlet, blocking APH heat transfer passages or causing blockage in SCR catalyst.
CaO reacts with SO2 and SO3 from flue gas to form calcium sulfate:
CaO + SO2 + 1/2 O2 • CaSO4 [Equation 4]
CaO + SO3 • CaSO4 [Equation 5]
Some CaSO4 is deposited on boiler tubes, causing low-temperature fouling, and some on the APH surfaces, where it is hydrated. The reaction described by [5] is mostly responsible for formation of calcium sulfate in APHs.
At lower temperatures (typical of the APH CE and HE layers) calcium sulfate is hydrated to form gypsum (calcium sulfate hydrate) or plaster of Paris (calcium sulfate hemi-hydrate):
CaSO4 + 2H2O • CaSO4 • 2H2O (Gypsum) [Equation 6]
CaSO4 + 1/2 H2O • CaSO4 • 1/2 H2O (Plaster of Paris) [Equation 7]
The water required for hydration comes from the flue gas or from the steam-blown sootblowers. Therefore, gypsum more likely formed in the CE layer, while plaster of Paris more likely formed in HE layer, where less H2O is available and temperatures are higher.
Pozzolanic (cement) reaction is also possible where hydrate lime reacts with clay from the coal (SiO2 Al2O3) to form portlandlite:
CaO + H2O • Ca(OH)2 [Equation 8]
Ca(OH)2 + SiO2 • Al2O3 • CaO • SiO2 • Al2O3 • 2H2O [Equation 9]
Once hardened, these deposits are very hard, or impossible, to remove. The APH pressure drop increases and, eventually, APH baskets need to be replaced.
The existence of these compounds was confirmed by advanced analysis of APH deposits. The best results are achieved by analyzing a deposit that is still attached to the metal surface. X-ray diffraction (XRD) analysis, followed by energy dispersive spectrometer (EDS) mapping, provides information on compounds present in the deposit (CaSO4, SiO2, Al2O3) and morphology of the deposit, which can be extremely complex, consisting of several deposition layers and sub-layers.
Options for Controlling APH Fouling and Corrosion
Fouling and corrosion of APHs can have significant impact on unit performance and availability. Net unit heat rate is affected by the higher temperature of the flue gas leaving the APH, which is caused by reduced heat transfer (fouling of the heat transfer surfaces) or loss of heat transfer surface due to corrosion and by higher forced draft (FD) and induced draft (ID) fan power requirements due to increased pressure drop (also caused by fouling). Unit availability is affected by scheduled and unscheduled APH washes, which are needed to remove fouling deposits and restore, if possible, pressure drop across the APH.
The options available for controlling APH fouling and corrosion of heat transfer surfaces in the cold end and intermediate layers include:
- Sootblowing
- Water wash
- Increased cold end temperature
- Reduction of SO3 concentration in the flue gas
- APH modifications
- APH replacement
Sootblowing. APHs are equipped with sootblowers located at the cold and hot ends of the APH. Steam or compressed air is used as a sootblowing medium. For bituminous coals, field experience shows that sootblowers are relatively effective in removing fouling deposits from the APH cold end that are formed by the deposition of sulfuric acid. In case of heavier fouling, water washes are needed to maintain the APH pressure drop.
Effectiveness of the HE sootblowers is often debated, and many plants have removed them. If a plant is considering injection of sorbents for SO3 or Hg reduction, it is recommended that the HE sootblowers be left in place, because they might be needed to maintain cleanliness of the APH HE once sorbent injection starts.
For low-rank fuels, deposits forming in the CE and HE layers of the APH are mostly calcium sulfate–based [Equations 7 to 9]. If allowed to harden, these deposits are very hard or impossible to remove. Therefore, if plant-specific data show benefits of sootblowing in decreasing the rate of the APH pressure drop increase, an aggressive sootblowing might be needed. In cases where CE sootblowing is not very effective, additional moisture might be aggravating the CE and HE fouling by providing additional moisture for hydration of calcium sulfate. Use of compressed air might be beneficial in these cases.
There is debate about whether sootblowing helps or hurts when low-rank fuels are fired. In some cases, sootblower jets do not remove fouling deposits; instead, they push them back, deeper into the CE layer, as presented in Figure 17.

17. Effect of sootblowing on fouling deposits formed when using PRB coal. Source: ERC
As shown in Figure 17, sootblowing jets penetrate about 9 to 10 inches into the CE layer (which is 12 inches deep) and remove most of the deposits from the surface. However, these deposits are pushed deeper into the CE layer of the APH and, in this case, the effectiveness of sootblowing on APH pressure drop is debatable.
The high-energy, dual-media sootblowers use smaller-diameter nozzles and higher pressure to increase jet penetration. High-pressure (HP) steam or HP water can be used (Figure 18). HP water is used for online or, most often, for off-line APH water washes.

18. High-energy, dual-media sootblowers. Source: ERC
Water Wash. APH water-washing might be needed when firing medium- and high-sulfur bituminous coals, where APH sootblowers cannot keep up with fouling rates. With low-rank coals, water washes are often needed to remove calcium sulfate–based deposits from the CE and HE layers of the APH.
In case of fouling caused by ABS deposits, frequent APH water washes are usually needed. This is because the ABS deposits in the HE layer of the APH, approximately 2 to 3 feet from the CE of the APH, and sootblowing jets either cannot penetrate to this location or their energy is insufficient to remove soft and sticky ABS deposits.
Although water-washing itself may take 6 to 14 hours, the overall process, including shutting down and cooling the unit, may bring the total outage to about 30 hours. Also, after water-washing, the APH has to be dried by rotating it through the combustion air stream (with the flue gas duct shut or isolated) to prevent fouling of wet surfaces by fly ash. APH drying might increase overall water-washing time beyond 30 hours.
For boilers equipped with two APHs and the capability to isolate them, online APH washing might be possible. This is accomplished by reducing the unit load to 50% to 60%, isolating one APH, and washing at reduced temperature. Online water-washing is performed using mid-pressure (90 psi to 150 psi, or higher) water and permanently installed wash pipes or dual-media sootblowers. For boilers with one APH or with no capability to isolate an individual APH, the unit has to be shut down for washing. Off-line water washing is usually performed by contractors using high-pressure (4,000 psi) water.
When dealing with ABS fouling, water-washing is often the only effective means of APH cleaning. However, field experience has shown that overreliance on water-washing may increase APH surface corrosion, which, in turn, will increase fouling rates.
Increase Cold End Temperature. The traditional utility guidelines for CE temperature control specify the APH CE average temperature (CEAT) as a function of coal sulfur content. This approach greatly oversimplifies the problem. In addition to sulfur content, there are many important unit-specific design and operating parameters that affect the H2SO4 concentration in the flue gas and acid deposition rate in the CE of the APH. There are significant economic penalties for operating with CEAT that is too high (increased stack losses and higher net unit heat rate) or not high enough (increased APH fouling and pressure drop, higher FD and ID fan power, lower power output or unit derate, higher maintenance, and reduced unit availability). Therefore, optimal CEAT has to be determined on a unit-specific basis.
One of traditionally used options for increasing CEAT involves increasing the combustion air temperature in the APH by preheating it in a steam air heater (SAH). Steam for the SAH is extracted from the steam turbine cycle. The second traditional option involves bypassing a fraction of combustion air around the APH. This lowers the capacity rate ratio (CR) of the APH and, as discussed earlier, increases the temperature of the flue gas leaving the APH.
An increase in temperature of the combustion air at the APH inlet increases the temperature of the heat transfer matrix. This increase is highest at the CE of the APH, while metal temperatures at the HE of the APH are affected very little by an increase in the inlet air temperature. Figure 19 shows the effect of inlet air temperature on axial (longitudinal) variation of minimum metal temperature and the onset of acid deposition. As the inlet air temperature is increased from 80F to 200F, metal temperatures at the APH CE increase by approximately 100F. With higher metal temperatures, the onset of acid deposition is shifted closer to the CE of the APH. For the example shown in Figure 19, increasing the inlet air temperature from 80F to 160F shifts the onset of acid deposition from the HE layer (1 foot, 4 inches from the CE) to the CE layer (41/2 inches from the CE), shrinking the size of the acid deposition zone by approximately four times.
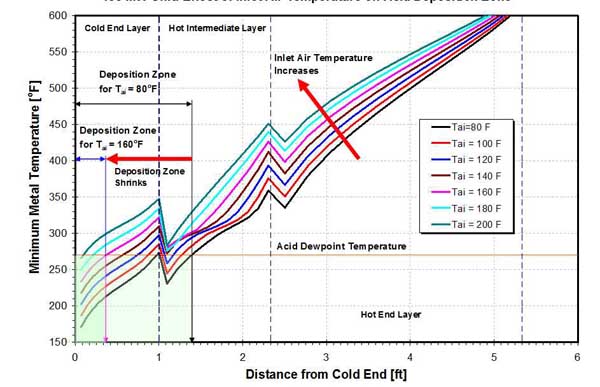
19. Effect of inlet air temperature on acid deposition zone on a 458-MW unit. Source: ERC
Also, as the temperature of the inlet air is increased, the temperature of flue gas (Tgo) and combustion air (Tao) leaving the APH increases (Figure 20). The increase in Tgo is approximately two times larger than the increase in Tao. On average, a 1-degree F increase in Tai result is approximately a 0.5-degree F increase in Tgo and a 0.25-degree F increase in Tao.
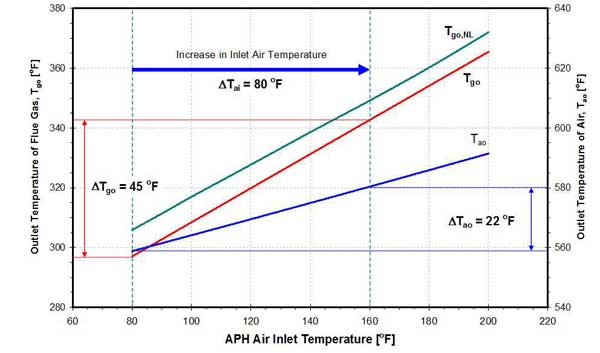
20. Effect of inlet air temperature on outlet air and flue gas temperatures on a 458-MW unit. Source: ERC
As discussed earlier, the temperature of inlet air is raised by increasing steam extraction from the steam turbine. The heat rate penalty, presented in Figure 21, is a total penalty due to the higher temperature of flue gas leaving the APH (higher stack gas loss) and higher steam extraction flow.
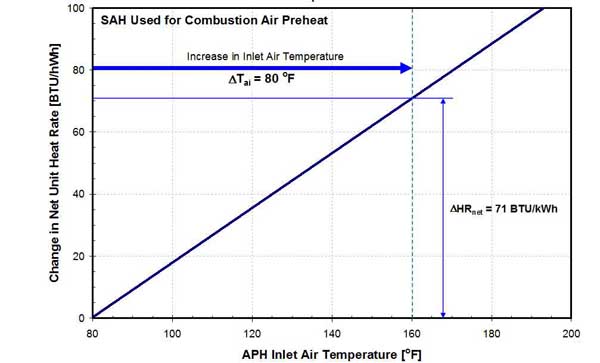
21. Effect of inlet air temperature on net unit heat rate on a 458-MW unit. Source: ERC
In the absence of a real-time indication of matrix temperature, plant engineers rely on the flue gas temperature, measured at the APH outlet, and CEAT guidelines to maintain an acceptable level of acid condensation in the APH. As shown in Figure 22, the difference between the flue gas temperature at the APH outlet and metal matrix temperature can be large. For this example, the average metal temperature at the exit of the APH is, on average, approximately 120F lower compared to the measured flue gas temperature, and it is below the acid dewpoint temperature except for Tai >180F. The metal temperature at the inlet to the APH CE layer is approximately 100F higher compared with the metal temperature at the outlet of the CE layer. A 12-inch-deep CE layer composed of notched-flat (NF) heat transfer surface was assumed in this example. The analysis can also be extended to the ABS deposition zone.
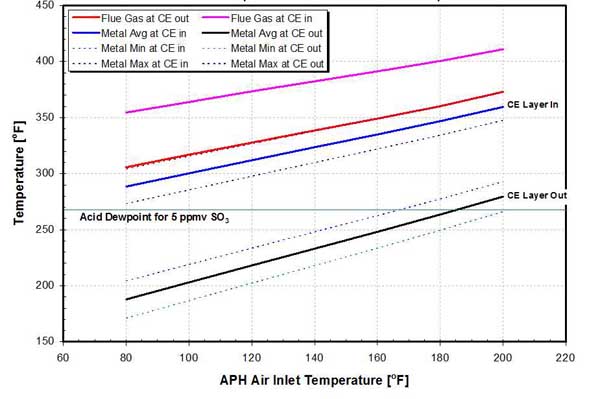
22. Effect of inlet air temperature on metal temperatures on a 458-MW unit. Source: ERC
The other option for increasing the flue gas temperature at the APH outlet and raising CEAT involves bypassing a fraction of the combustion air around the APH. The cold air bypass option was analyzed and compared with the air preheat option. The results are presented in Figures 23 and 24.
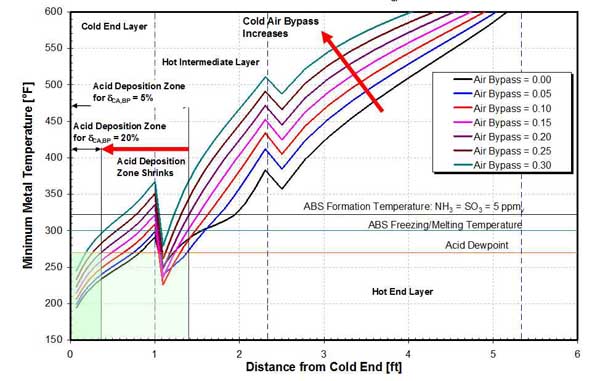
23. Effect of cold air bypass on acid deposition zone on a 458-MW unit with an ambient inlet temperature of 110F. Source: ERC
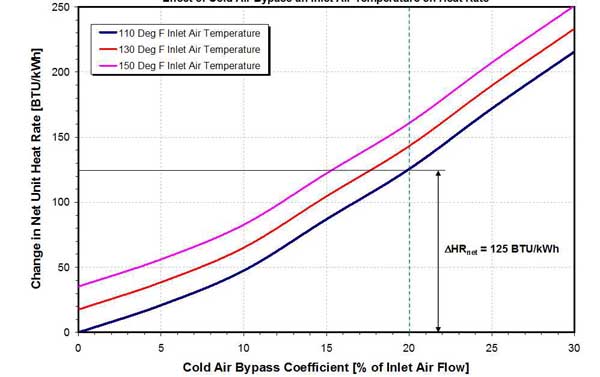
24. Effect of cold air bypass and inlet air temperature on heat rate penalty on a 458-MW unit. Source: ERC
As the amount of air bypassing the APH is increased, the capacity rate ratio of the APH is decreased. This decrease in CR increases the temperature of the flue gas leaving the APH, which, in turn, leads to higher temperature of the metal matrix. The effect of the cold air bypass on axial (longitudinal) variation of minimum metal temperature, and the onset of acid deposition, is presented in Figure 23. As the cold air bypass is increased from 0% to 30%, metal temperatures at the APH CE increase by approximately 50F. With higher metal temperatures, the onset of acid deposition is shifted closer to the CE of the APH. For the example shown in Figure 23, increasing the cold air bypass from 0% to 20% shifts the onset of acid deposition from the HE layer (approximately 1 foot, 4 inches from the CE) to the CE layer (4 1/2 inches from the CE), shrinking the size of the acid deposition zone by a factor of four.
The heat rate penalty incurred by cold air bypass is presented in Figure 24. The results show that the heat rate penalty caused by cold air bypass is substantially higher than the penalty for preheating inlet air (see Figure 21). This is because of higher-temperature flue gas (higher stack gas loss) and lower-temperature hot combustion air to the boiler (lower heat credit to the boiler). The amount of cold air bypass can be decreased by preheating the combustion air. In general, preheating of combustion air is a preferred option because of the lower heat rate penalty and higher metal temperatures in the CE of the APH (see Figures 19 and 23).
Advanced Air Preheating
Another possibility for increasing the temperature of combustion air entering the APH involves using heat recovered from the flue gas. (See Part I of "Flue Gas Heat Recovery in Power Plants" for a discussion of using low-temperature heat for feedwater and combustion air preheating.) The analysis involved two cases: one in which combustion air was preheated in a steam air heater (SAH) using steam extracted from the steam turbine and another in which the same amount of heat was provided by heat recovered from the flue gas in the flue gas cooler (FGC).
The analysis was performed where the amount of heat recovered from the flue gas was varied to determine the effect on the temperature of the flue gas leaving the APH and net unit heat rate. The results are presented in Figures 25 to 27. The system configuration, including the combustion air heater (AH) and FGC, is presented in Figure 25.
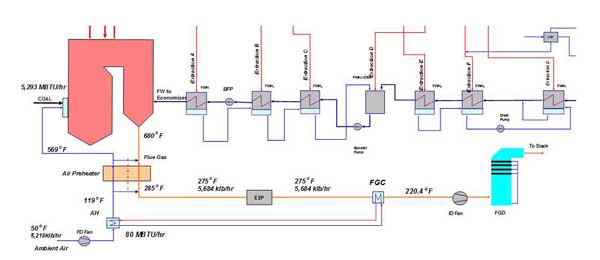
25. System schematic illustrating advanced air preheating. Source: ERC
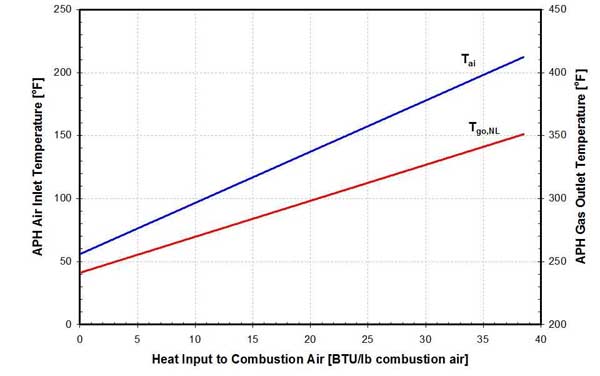
26. Effect of advanced air preheating on air and flue gas temperatures on a 650-MW unit burning washed Illinois coal. Source: ERC
27. Effect of advanced air preheating on net unit heat rate on a 650-MW unit burning washed Illinois coal. Source: ERC
As the amount of heat recovered from the flue gas in the FGC increases, the temperature of the combustion air entering the APH increases (Figure 26). Higher inlet air temperature results in higher-temperature flue gas leaving the APH. The flue gas temperature, corrected for no leakage (Tgo,NL), is presented in Figure 26.
The results show that using heat recovered from the flue gas downstream of the APH makes it possible to increase the temperature of the combustion air entering the APH and the flue gas leaving the APH. More importantly, these increases can be accomplished without negatively affecting net unit heat rate, as is the case when the SAH and cold air bypass are employed. In case of advanced air preheating, the net unit heat rate is improved as the inlet air temperature is increased (Figure 27). This is because heat from the flue gas stream is recovered and used for air preheating, while in traditionally used approaches, the higher temperature of the flue gas leaving the APH increases stack loss and net unit heat rate (see Figures 21 and 24).
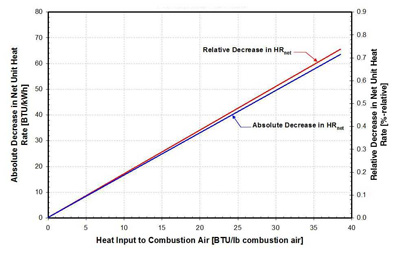
27. Effect of advanced air preheating on net unit heat rate on a 650-MW unit burning washed Illinois coal. Source: ERC
The other methods for increasing the temperature of flue gas leaving the APH and CEAT involve reducing the rotational speed of the APH and increasing the flue gas temperature into the APH. Both are ineffective in increasing CEAT.
Reduction of SO3 Concentration in Flue Gas. Although the extent and severity of corrosion varies from plant to plant, the resulting damage is greatest to ductwork, the ESP, and the blades and housing of ID fans. Repairing corrosion damage at every opportunity is important because corrosion leads to costly increases in air in-leakage. It is not unusual for a utility to spend several million dollars every few years to repair corrosion damage and/or replace damaged components. Although the costs of corrosion damage are very site-specific, the financial benefit derived from the reduction of SO3 concentration in the flue gas and the subsequent reduction of corrosion in back-end equipment could be significant.
Because SO3 will not condense until cooled to its dew point, the potential for corrosion can be reduced by reducing the concentration of SO3 at the APH inlet. The lower the SO3 concentration at the air heater inlet, the lower the sulfuric acid dew point and the lower the potential for corrosion of back-end equipment. With the unit firing bituminous coal; an 8% flue gas concentration of H2O; and SO3 concentrations of 60, 30, and 3 ppmv, the corresponding dew points are 309F, 296F, and 254F, respectively.
If the SO3 level at the entrance to the APH is reduced, the APH can be operated with a lower outlet gas temperature, allowing the APH to recover additional heat from the flue gas without increasing the downstream corrosion potential.
The potential for ABS formation and fouling also is greatly reduced. This can reduce or eliminate the need for unit derates or outages for water-washing to remove ABS deposits. Lower acid or ABS deposition rates would also extend basket life because of reduced corrosion, sootblowing, and water-washing frequency.
Reducing SO3 levels and corrosion potential also reduces cost by eliminating the need to modify and coat air heater baskets to protect them and make them easier to clean. It is reasonably common for power plants, when they add an SCR system, to modify the physical configuration of the air heater to allow better cleaning access to the areas where ABS, a sticky solid, is prone to form and deposit.
The most popular procedures for reducing SO3 concentration in the flue gas stream involve injection of a sorbent into the fuel stream, into the furnace, or into the flue gas stream. When added to the fuel stream, limestone represents approximately 0.2% to 0.8% of the coal feed. Alkali (limestone) injection also helps mitigate SCR catalyst poisoning by arsenic.
The sorbent reacts with SO3 in the flue gas to form a solid compound that can be removed in the plant’s particulate collection device—an electrostatic precipitator or fabric filter. Also, the compounds formed should not deposit on the APH heat transfer surfaces; otherwise, one fouling mechanism is replaced with another.
The amount of SO3 removed and the amount and cost of reagent (sorbent) required for the reduction for a unit operating at a 30 ppmv SO3 would be one-half that for one running at 60 ppmv SO3. Therefore, the lower the concentration of SO3 in the target stream, the greater the savings potential. In other words, for units burning low-sulfur coal, SO3 reduction is easier to achieve. For example, removing 95% of the SO3 from a flue gas stream containing 10 ppmv SO3 would decrease acid dewpoint by 50F (from 275F to 225F); at the same removal rate on a 60 ppmv SO3 stream, the dew point would be decreased by 55F (from 309F to 254F).
Assuming that the APH flue gas outlet temperature can be reduced by an amount equivalent to the decrease in the acid dewpoint, and that a change of 25F in that temperature equates to a 1% (relative) change in unit heat rate (Figure 1), it would result in a 2% improvement in net unit heat rate for a 30 ppmv SO3 stream and a 2.2% improvement for the 60 ppmv SO3 stream. It has to be noted that an existing APH would have to be modified to allow transfer of additional heat from the flue gas to combustion air.
SBS Injection
Sodium bisulfite (SBS) injection technology involves the injection of a solution of sodium bisulfite and/or sodium sulfite into the flue gas for the selective removal of SO3. The technology is patented by Codan Development LLC and is being offered by Codan with URS Corp. providing design, detailed engineering, procurement, installation, start-up, and operations assistance services.
The SBS solution is injected into the flue gas stream as a fine mist through dual fluid nozzles. The moisture evaporates within a few feet of the injection point. The dilution water must be low in calcium to avoid having scale form in piping and spray nozzles. Injection system controls maintain a constant feed rate of solution to the nozzles to achieve consistent droplet atomization. The amount of concentrated reagent in this stream is also controlled to follow the flue gas SO3 concentration and unit load. The solution is atomized by compressed air into fine droplets and injected into the flue gas stream. The droplets are converted to very small dry particles of high surface area within a few feet of the injection point. The particles then are removed with other flue gas particulates by either an ESP or a fabric filter.
Though some important reactions occur within that very short time in the liquid phase, the majority of the SO3 removal occurs as a solid phase reaction with the dried particles. The particles formed are small, and due to the "popcorn" effect of being formed as SO2 is off-gassed, have a very high surface area and are, therefore, quite reactive. The dry reaction products are removed from the gas stream with the particulates, normally in an ESP or a fabric filter.
SBS can be injected upstream or downstream of the APH. The preferred location for injection is immediately downstream of the SCR (or the economizer, if there is no SCR). From a practical view, the location upstream of the APH is most desirable because the reaction proceeds faster at the higher temperature, and less drying time is required. Most importantly, however, removal of SO3 upstream of the APH provides additional benefits, as previously discussed. Downstream of the SBS injection point, it is necessary to have a minimum section of duct free of any internal obstructions, usually 8 to 10 feet, to allow adequate drying time before the solids contact any surfaces to prevent solids deposition problems.
SO3 removal efficiency ranges from 90% to over 98%. However, the effectiveness of the SBS injection technology (or any other sorbent injection technology) depends on good atomization of the injected solution, good dispersion of the liquid into the flue gas stream, the amount of reagent used, and the gas residence time. Although the first three issues can be dealt with by computational fluid dynamics (CFB) modeling and system design, retrofit projects often do not provide much space between the SCR system and the APH (shortening the residence time). Codan and URS are currently investigating whether SBS injection upstream of the SCR would have any adverse effects on the catalyst. Preliminary results are encouraging, and it is likely that SBS injection upstream of the SCR will be incorporated into future applications.
To date, SBS injection technology has been commercially installed on units totaling 8,500 MW of generating capacity, with units ranging in size from 287 MW to 860 MW, while SO3 concentrations range from 42 ppmv to 110 ppmv.
Trona and Lime Injection
Trona (a sodium-based product) and hydrated lime (a calcium-based product) are often used to reduce SO3 emissions. Mined trona, naturally occurring as a sodium sesquicarbonate (Na2CO3 • NaHCO3 • 2H2O), is converted into sodium carbonate and is also used for reduction of HCl, SO2, and mercury emissions. Although trona is more reactive than hydrated lime (due to its sodium base), and therefore less product is required for a given percentage removal, the optimal sorbent for a particular application will depend on factors including sorbent cost, transportation cost, equipment cost, and availability.
Trona and hydrated lime are usually delivered to a site via truck or rail car and off-loaded via pneumatic conveying to a storage silo. From the silo they are metered into a second pneumatic conveying system, where they are transported over moderate distances to a suitable injection location within the flue gas duct. The temperature of the flue gas at the injection point has to be higher than 275F. This conveying line, being of a relatively small diameter, can often be routed along existing structures, which reduces both material and installation costs. The pipe diameter is a function of the amount of sorbent being carried and the amount of conveying air used to transport the sorbent.
Removal efficiencies will vary according to the type of sorbent, sorbent injection rate, sorbent particle size, sorbent residence time, sorbent penetration and mixing with the flue gas, the temperature of the flue gas (275F to 800F, the higher the better), and the type of air pollution control (APC) equipment downstream. A particulate control system incorporating a fabric filter can typically achieve higher removal efficiencies than an ESP. However, depending upon the sorbent and quantity, an ESP may still be sufficient to obtain a desired acid gas (SO3 and SO2) reduction and maintain the existing level of particulate emissions.
The effect of trona injection on acid dewpoint temperature is presented in Figure 28. The plant, burning high-sulfur bituminous coal, was retrofitted with an SCR, which has increased the SO3 concentration in the flue gas upstream of the APH from 23 ppmv to 41 ppmv. The corresponding increase in acid dewpoint was 11F (from 293F to 304F). With the SCR in service, injection of trona has reduced SO3 concentration to 20 ppmv, depressing the acid dewpoint to 291F.
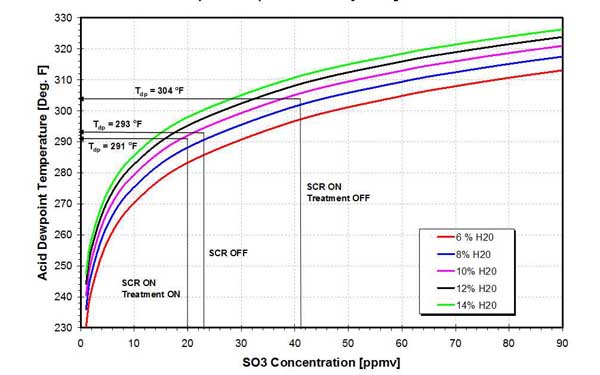
28. Effect of SCR operation and trona injection on SO3 concentration and acid dewpoint. Source: ERC
The effect of trona injection on the location and size of acid and ABS deposition zones is presented in Figures 29 and 30 for a two-layer Rothemuhle APH. ABS is formed when ammonia reacts with SO3 as flue gas cools in the APH. Ammonia enters the flue gas as slip from the SCR or SNCR system. SO3, at a concentration of 30 ppmv to 60 ppmv, can be a strong driver of ABS formation even if ammonia slip is low.
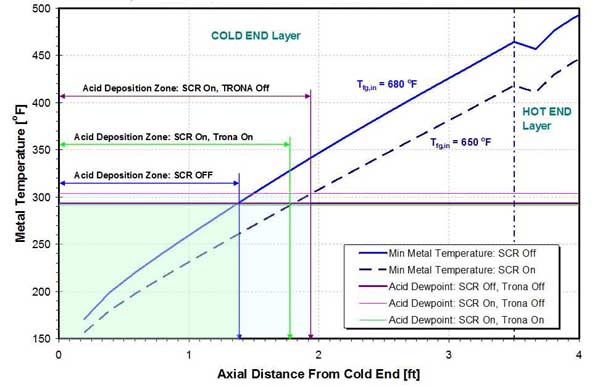
29. Effect of SCR operation and trona injection on acid deposition zone on a two-layer Rothemuhle APH. Source: ERC
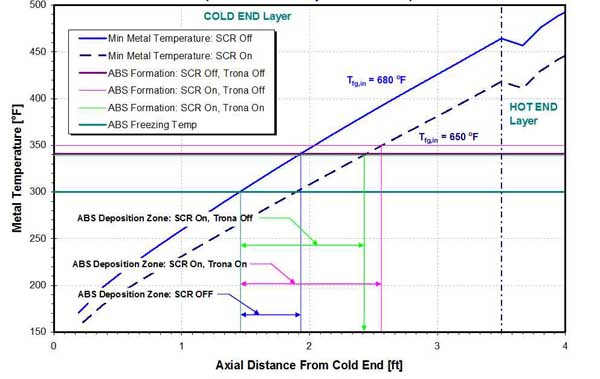
30. Effect of SCR operation and trona injection in ABS deposition zones in a two-layer Rothemuhle APH. Source: ERC
The catalysts used in the first wave of deployments of SCR systems in the U.S. were designed to have high activity so they could achieve high NOx removal efficiencies. However, high SO2 to SO3 conversions, in the 1.5% to 2.0% range, were not uncommon several years ago and were specified by original equipment manufacturers and architect/engineers. Over the past few years, much progress has been made in reducing the oxidation level to well below 0.5%.
In the example presented below, when the SCR is out of service (bypassed), the temperature of the flue gas at the APH inlet is 30F higher, compared with the SCR being in service. Also, the flow rate of the flue gas is higher, resulting in higher metal temperatures when the SCR is bypassed. The results from Figure 29 show that, with the SCR in service, the onset of acid deposition occurs earlier (farther from the APH CE) compared to the case where the SCR is off, resulting in larger acid deposition zone. The same is true for the ABS deposition zone (Figure 30).
Injection of trona depresses acid dewpoint and decreases the size of the acid and ABS deposition zones. However, in this example, the effect of trona injection on location and size of deposition zones is relatively limited, and an increase in the APH air inlet temperature is needed for significant reduction of the acid and ABS deposition zones. Calculations show that increasing the APH inlet air temperature from 100F to 160F will reduce deposition zones by a factor of two.
The results from this example should not be extrapolated to all APHs; a site-specific analysis is required to determine optimal operating conditions for each APH.
AEP pioneered the use of trona for SO3 mitigation at its General Gavin plant and then introduced its proprietary process across AEP’s entire bituminous coal-fired fleet where both SCR and FGD systems are in place. One of the leading suppliers of trona to the power industry is SOLVAir Products, part of Solvay Chemicals Inc.
Dominion Generation implemented a limestone sorbent injection system at its Chesterfield Station Unit 5 using the CleanStack technology jointly developed by the University of North Dakota’s Energy & Environmental Research Center, Marsulex Inc., and Alstom Power’s Air Preheater Co. (A complete description of that project is available here.) CleanStack promotes condensation of SO3 by injecting ultrafine particulate material (limestone, less than 4 microns in diameter) immediately upstream of the APH. The particle concentration provides nucleation sites for the condensation of sulfuric acid formed from SO3 and moisture in flue gas. The condensation process does not depend on the composition of the particles but only on their concentration and the particle-size distribution. By substantially increasing the concentration of small particles in fly ash (typically, from 1.5% to 4% by weight), the CleanStack process aids in SO3 removal. At Chesterfield Unit 5, injection of ultrafine limestone has reduced SO3 concentration in the flue gas by 50%. Based on its success at Unit 5, Dominion Generation has installed the Clean Stack system at Chesterfield Unit 6.
The common thread between the AEP and Dominion Generation projects, and many other successful SO3 mitigation projects across the industry, is finding the right sorbent to optimize SO3 reduction given the specific boiler and air-quality control system components in place at a plant.
ABS deposition also can be reduced by better control of reagent (ammonia or amine) injection. NH3 concentration has a significant effect on ABS formation temperature for NH3 <10 ppmv or 5 ppmv. The reduction in ammonia slip can be accomplished by periodic SCR tuning, by coordinating SCR/SNCR controls with the boiler controls, and by avoiding sudden load changes, especially load decreases. When unit load is suddenly decreased, it takes time for the SCR controls to react and decrease injection of the reagent. In the meantime, there is excess reagent in the flue gas, causing high ammonia slip and heavier APH fouling by the ABS.
APH Modifications
Modifications to the APH design to improve cleanliness of the heat transfer surfaces include heat transfer elements with closed flow channels, application of better materials, and a deep CE layer.
Heat Transfer Element. The heat transfer element is the heart of the APH, and its design is a key to the overall performance. Element profiles for regenerative APHs can be characterized as one of two general types: closed or open (Figure 31). Commercial regenerative heat transfer elements are composed of a pair of element sheets appropriately spaced to provide a flow passage between them. An example of a closed element profile is the notched flat 6-mm element (NF6), as shown in Figure 31. The element pair is formed by a series of notches that rest on an adjacent flat sheet with contact along the total flow length. They provide the necessary spacing and form discrete individual flow channels of fixed cross-sectional area along the flow length or element depth. There is no flow communication from one channel to the adjacent one.

31. APH heat transfer element design types. Source: ERC
The double undulated (DU) element, shown in Figure 31, is an example of an open-channel element, where the notches, which provide the required plate spacing, rest on a series of point contacts on the adjacent sheet. Flow can move across the element pair because there are openings between the two sheets along the flow length between point contacts.
Other variations of these basic types exist and vary among manufacturers, such as corrugated undulated (CU), flat notched crossed (FNC), or notched plate (NP). New advanced surface types such as UNU and DNF suggest improved performance over older types.
Although the choice of element profile for a given APH design is driven by many factors (such as heat transfer and pressure drop), from the standpoint of sootblowing effectiveness, closed-element profiles are preferred, because they contain the flow energy from the sootblower and thereby offer better removal of deposits compared to the open-element profiles. In an open-channel element, the sootblower energy can deflect (dissipate) to the sides and flow around the deposit.
Material Selection. The corrosive, dust-laden environment to which heating elements are exposed requires that material selection be an important design consideration. Historically, carbon steel and low-alloy corrosion-resistant (LACR) materials have been extensively used. The experience with ABS fouling and the increased propensity for acid attack on element surfaces have led to the use of enameled plates in the critical temperature zones of the APH. In Germany this use is widespread, resulting in lower deposition and corrosion rates. This has been corroborated in a test program in the U.S., as part of the Clean Coal Technology Program. Test results comparing carbon steel, LACR, and enameled elements indicated that the enameled plates have higher performance.
The test program, conducted at Southern Company’s Plant Crist, consisted of installing alternating elements of different materials in one sector of each of two APHs (APH A and APH B). Deposition and corrosion were characterized. Enameled elements exhibited about an order of magnitude less accumulation of deposits and corrosion loss compared with the carbon steel or LACR elements (Figures 32 and 33). It is estimated that these design modifications represent an incremental cost of 10% to 15% over a conventional design.
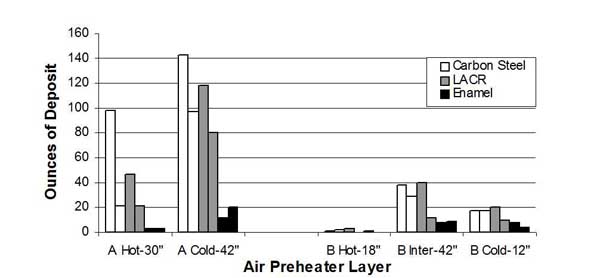
32. Effect of material selection on fouling of APH heat transfer elements. Source: ERC
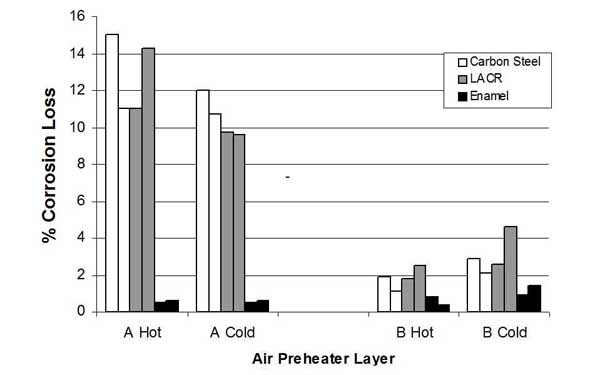
33. Effect of material selection on corrosion of APH heat transfer elements. Source: ERC
Deep Cold End Layer. The depth of the heating element layer and the use of single versus split designs impacts the propensity for and the severity of ABS deposition. First, the lengths of the three commonly used layers (hot, intermediate, cold) of the APH affect the axial (longitudinal) temperature profile. This temperature profile is one of the major determinants of whether and where deposition will occur. In general, shallower HE layer depths have a higher risk of fouling.
Second, split layers cause a physical disturbance in the flow direction in the form of a gap within the layers. With a three-layer design, heavy ABS deposits can form on the face of the HE layer. The use of a continuous-element sheet (deep CE layer) has several advantages. Most important is the fact that there is no break in the element continuity where deposits can occur. Experience has shown that when sootblowing is used, deposits will accumulate on the downstream edge of the layer. The aerodynamics of the sootblowing process in the gap region between the CE and HE layers is such that eddies and backflow swirls form and result in poor cleaning performance of a sootblower jet.
Traditional APH designs have used 12-inch CE and 18-inch HE layers. As shown in Figure 34, the split between the CE and HE layers can be located within the ABS formation zone. If so, heavy fouling of the HE layer will occur, which sootblowers located at the CE of the APH will not be able to remove. To overcome or mitigate this problem, a two-layer APH design, involving a deep CE layer (up to 48 inches), was developed.
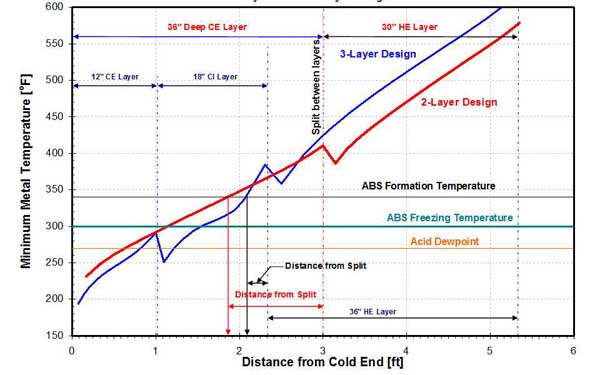
34. Two-layer versus three-layer APH design on a 458-MW unit. Source: ERC
A comparison of temperature profiles for a three- and two-layer APH design is presented in Figure 34. The ABS and sulfuric acid deposition zones are also shown. With a 36-inch-deep CE layer, the ABS and sulfuric acid deposition are contained in a single, continuous layer. This allows for better cleaning, especially if enameled heat transfer surfaces with closed channel elements are used.
To further illustrate this design change, a typical arrangement of an APH supplied about 15 years ago is compared with the modern configuration. The modern APH configuration is reduced in depth by 21 inches and in weight by about 25%. Pressure drop is 20% lower.
The modification’s impact on the APH pressure drop must also be considered. If the elimination of ABS formation reduces fouling, the fan capacity that had previously been needed to compensate for the pressure drop increases between planned outages would now be available to offset the higher pressure drop requirements of basket designs with a higher surface area or higher heat transfer coefficient.
Complete APH Replacement. A complete replacement of the APH is the most expensive option and is warranted in situations where structural integrity of the old APH is compromised, all baskets need to be replaced, and air leakage is very high. For example, this would be a situation where an old Rothemuhle-type of APH with air leakage exceeding 30% and corroded and damaged baskets is replaced by a modern Ljungstrom type APH with modern seals and high-efficiency heating elements.
More to Come
In Part III, we’ll look at options for improving the performance of boiler auxiliaries by reducing the auxiliary power they use.
—Nenad Sarunac (ns01@lehigh.edu) is principal research engineer and associate director at the Energy Research Center, Lehigh University. The Illinois Clean Coal Institute funded a portion of this work.