Solid oxide fuel cells (SOFCs), which oxidize a fuel to produce electricity, have received much attention of late for the technology’s myriad benefits, including high efficiency, long-term stability, fuel flexibility, and low carbon emissions—all at a relatively low cost. They have been used to date in a number of applications, including as auxiliary power units in vehicles and for stationary power generation with capacities ranging from 100 W to 2 MW. Their high operating temperatures—between about 1,100F and 1,800F—make them suitable for applications with heat engine energy recovery devices or to increase overall fuel efficiency at combined heat and power plants. In May and June, SOFC researchers announced three significant achievements.
Reaching for 60% Efficiency
In a study published in May’s issue of the Journal of Power Sources, scientists at the U.S. Department of Energy’s Pacific Northwest National Laboratory (PNNL) say they have developed a small, 2-kW SOFC system that achieves 57% efficiency—much higher than the 30% to 50% efficiencies conventionally reported for systems of similar size—that could offer a “viable option of highly efficient, localized power generation,” according to Vincent Sprenkle, a coauthor of the study and chief engineer of PNNL’s SOFC development program.
As PNNL explains, SOFCs—and other fuel cells—are like batteries in that they use anodes, cathodes, and electrolytes to produce power. But unlike batteries, which stop working when they use up their reactive materials, fuel cells can continuously make electricity if they have a constant fuel supply. SOCFs offer the added advantage of being able to run on a variety of fuels, including natural gas, biogas, hydrogen, and liquid fuels.
SOFCs are typically made of up ceramic materials that form three layers: anode, cathode, and electrolyte. As air is pumped up against the outer layer—the cathode—oxygen from the air becomes a negatively charged ion, O2 –, where the cathode and inner electrolyte meet. The ion moves through the electrolyte to reach the final layer—the anode—where it reacts with a fuel and produces electricity and byproducts of carbon dioxide and steam.
PNNL scientists claim that SOFCs can convert up to 60% of the chemical energy in fuel into electricity, making them much more efficient than the combustion engines of portable generators, which only convert 18%. Rather than focusing on larger systems, Sprenkle and his PNNL colleagues have centered their research on smaller systems, which they say could be placed closer to power users and nix transmission losses. The pilot system they built and tested generates about 2 kW of electricity and has been designed to be scaled up to between 100 kW and 250 kW.
The lab-built SOFC system uses external steam reforming (Figure 1), a process that mixes steam with methane. The reaction of these two components forms carbon monoxide and hydrogen, intermediate products that then react with the oxygen at the fuel cell’s anode. Although steam reforming has been used with fuel cells before, external steam reforming, which completes the initial reactions between steam and fuel outside the fuel cell, avoids compromising the ceramic layers with the use of uneven temperatures.
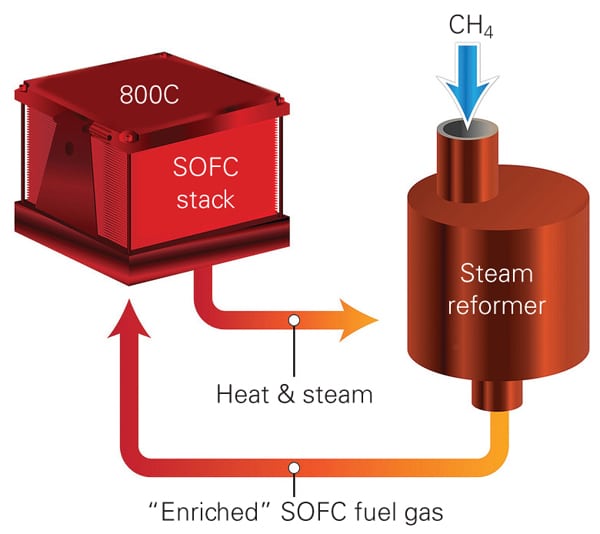 |
| 1. New channel of innovation. A new small-scale solid oxide fuel cell (SOFC) system developed by Pacific Northwest National Laboratory that uses methane as fuel promises efficiency of up to 57%, much higher than the 30% to 50% reported for other systems its size. The SOFC uses microchannel technology and two unusual processes: external steam reforming and fuel recycling. Source: PNNL |
The process instead uses a heat exchanger to move hot gas expelled as a byproduct of the reaction inside the fuel cell to warm up cooler incoming gas to temperatures needed for the reaction taking place inside the fuel cell.
PNNL’s SOFC system is more efficient, the scientists say, because it uses a lab-developed microchannel technology: Instead of having just one wall that separates the two gases, PNNL’s microchannel heat exchangers have multiple walls created by a series of tiny looping channels that are narrower than a paper clip and which increase the surface area, allowing more heat to be transferred. Little additional pressure is needed to move the gas through the looping channels. Efficiency is also boosted because the system uses the exhaust (made up of steam and heat byproducts coming from the anode) to maintain the steam-reforming process.
PNNL test results show that the system’s net efficiency ranges from 48.2% at 2.2 kW to 56.6% at 1.7 kW, and the team “calculates they could raise the system’s efficiency to 60% with a few more adjustments.” Yet, as Sprenkle concedes, “There still are significant efforts required to reduce the overall cost to a point where it is economical for distributed generation applications.”
Generating Power Without Fuel
In June, material scientists at Harvard University said they had demonstrated an SOFC that converts hydrogen into electricity that can also store electrochemical energy like a battery—meaning that the fuel cell can continue to produce power for a short time after its fuel has run out.
“This thin-film SOFC takes advantage of recent advances in low-temperature operation to incorporate a new and more versatile material,” explained principal investigator Shriram Ramanathan, associate professor of materials science at the Harvard School of Engineering and Applied Sciences. “Vanadium oxide (VOx) at the anode [as opposed to the platinum typically used] behaves as a multifunctional material, allowing the fuel cell to both generate and store energy.”
The researchers’ finding, published in the June issue of the journal Nano Letters, specifies that compared to a platinum-anode SOFC that generates power for just 15 seconds after fuel runs out and before the electrochemical reaction peters out, the new SOFC uses a bilayer of platinum and VOx for the anode, allowing the cell to continue operating up to 14 times as long (3 minutes, 30 seconds, at a current density of 0.2 mA/cm2). The early result is only a proof of concept, they say, though they predict that future improvements to the composition of the anode could further extend the cell’s life span. A more advanced fuel cell available for applications testing in micro-air vehicles (unmanned aerial vehicles), for example, could be available within two years, the researchers claim.
A Triple Combined Cycle Power Generation System
Mitsubishi Heavy Industries (MHI) said it would develop basic technologies for a triple combined cycle power generation system integrating SOFCs and a gas turbine combined cycle (GTCC) power generation system. The two-year study announced this June to integrate existing SOFC technology with a GTCC system focuses on generating power at three stages: the fuel cell, gas turbine, and steam turbine (Figure 2). It could result in what the Japanese firm calls a “fuel cell combined cycle (FCCC) system” that is expected to “achieve the world’s highest power generation efficiency exceeding 70% (lower heating value, LHV) for several hundred MW-class power generation and over 60% (LHV) efficiency for several tens MW class power generation.”
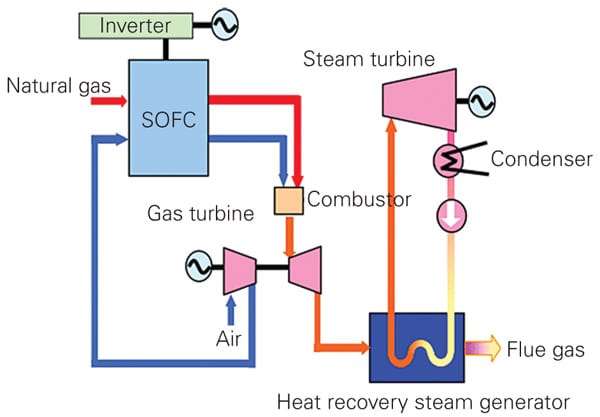 |
| 2. Triplets. Under the umbrella of the New Energy and Industrial Technology Development Organization, Mitsubishi Heavy Industries said it would begin developing basic technologies for a triple combined cycle power generation system integrating solid oxide fuel cells (SOFC) and a gas turbine combined cycle (GTCC) power generation system. As this conceptual drawing shows, the system would place the SOFC system before the GTCC system. Source: MHI |
As part of the study, MHI plans to develop the basic technologies needed to combine SOFC and GTCC systems. It will test the SOFC’s durability under pressures of up to 3.0 megapascal gauge 2 and study issues affecting conversion of the gas turbine and combustor development. The study will result in a demonstration of the FCCC and, ultimately, commercialization.
If it is successful, the new technology could become a “revolutionary, epochal” achievement that would raise the power generation efficiency of existing natural gas–fired systems by 10% to 20%.
—Sonal Patel is POWER’s senior writer.