Advanced SCR Catalysts Tune Oxidized Mercury Removal
Catalysts used in selective catalytic reduction (SCR) systems in utility boilers provide high NOx removal efficiencies that routinely exceed 90%. A major co-benefit of applying SCR to coal-fired power plants is that the SCR catalyst also oxidizes the vapor phase mercury from an elemental form to a soluble ionic form, which can be readily captured in a downstream flue gas desulfurization process. Mitsubishi Heavy Industries and Cormetech have developed an advanced SCR catalyst technology with high mercury oxidation activity capable of achieving 95% oxidized mercury over a wide range of operating conditions.
While U.S. mercury emission control rules are stalled at the federal level, many individual states have filled the gap with a patchwork of rules and requirements. It’s reasonable to expect that the Environmental Protection Agency (EPA) will eventually develop mercury rules that will pass judicial scrutiny and survive the inevitable series of lawsuits that follow (see sidebar). In the meantime, the important research and development work required to accurately quantify the co-benefits of air quality control system components continues.
A recent article in POWER (“Determining AQCS Mercury Removal Co-Benefits,” July 2010) discussed Southern Company’s extensive test program to determine the combined mercury removal co-benefits possible with the electrostatic precipitator (ESP), selective catalytic reduction (SCR), and wet flue gas desulfurization (FGD) technology combination. In this article, we explore another approach to maximizing mercury co-benefits: using an advanced SCR catalyst tuned to maximize mercury oxidation for a particular set of operating conditions.
Mercury Oxidation Chemistry
Coal combustion, most notably in utility boilers, has been targeted as a major source of mercury emissions. Mercury levels in coals mined in the U.S. typically range from 0.05 to 0.2 ppm. In the high-temperature regions of coal-fired boilers, mercury in coal is volatilized and converted to its elemental metallic form (Hg0). A complex series of reactions occurs as the flue gas is cooled, converting Hg0 to oxidized (ionic) mercury (Hg2+) and/or mercury compounds that are solid-phase (particle bound) (Hgp).
The partitioning of mercury into its three forms (Hg0, Hg2+, and Hgp) is referred to as mercury speciation, and the degree to which speciation occurs can substantially affect mercury control approaches. Mercuric chloride (HgCl2 or Hg2+) compounds are soluble and can be captured in FGD systems used for sulfur dioxide (SO2) removal. Hgp compounds or Hg compounds adsorbed onto the surface of other particles can be captured to varying degrees using particulate matter control devices such as fabric filters or ESPs. This process may be facilitated by use of additives such as activated carbon. The emissions control device capture points for oxidized and particle-bound mercury are shown schematically in Figure 1.
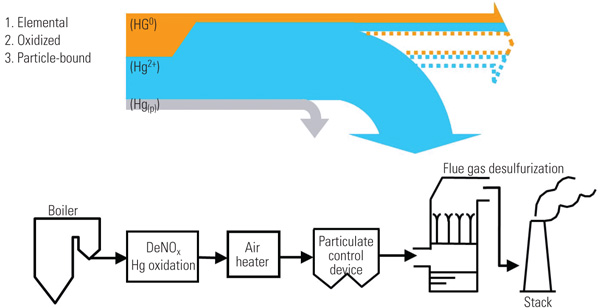 |
| 1. Triple play. Mercury capture points for the three forms of mercury in flue gas are illustrated. The gray arrow represents the particulate mercury removed in the particulate control device. The blue arrow represents the oxidized mercury that is removed in the flue gas desulfurization system. At the stack, the “total” arrow (the sum of the solid blue, solid orange, dotted blue, and dotted orange lines) represents the amount of mercury that comes out of the stack without a selective catalytic reduction and flue gas desulfurization system present. The solid blue plus the solid gold lines denote the smaller portion of elemental and oxidized mercury that leaves the stack because the SCR and FGD are present. Source: Cormetech Inc. |
It is also well known that catalysts used for selective catalytic reduction of NOx compounds can exhibit the co-benefit of promoting mercury oxidation. In addition to reacting with NOx compounds to form nitrogen and water, titania-based SCR catalysts containing oxides of vanadium and molybdenum or tungsten have been shown to be effective in oxidizing elemental mercury to its Hg2+ form. The SCR process and its key reactions are shown in Figure 2.
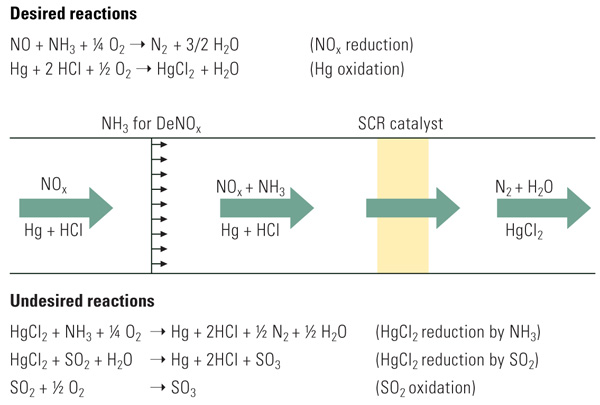 |
| 2. SCR process schematic and key reactions. Source: Cormetech Inc. |
Key Performance Parameters
A number of factors are known to affect the degree of oxidation of elemental mercury. Key factors include halogen content (for example, chlorine, and bromine), temperature, reductants (such as ammonia [NH3] and SO2), catalyst chemistry, and catalyst age, as well as the expected flue gas constituents from coal combustion.
Halogen Content. Halogen content in the fuel and flue gas is an important driver for achieving mercury oxidation through the SCR catalyst. Chlorine levels in bituminous coals such as Illinois Basin coal exceed 500 ppm, resulting in hydrocholoric acid (HCl) levels in the flue gas of greater than 35 ppm, thus helping to drive greater formation of oxidized mercury through the SCR. For subbituminous coals such as Powder River Basin coal, chlorine levels are typically <100 ppm, resulting in HCl levels in the flue gas of <10 ppm. This low level of halogen in the flue gas can limit the SCR performance for oxidizing mercury, and this presents one of the challenges to catalyst manufacturers. Methods to enhance performance include improved catalysts that can be used exclusively or in combination with halogen additives.
Temperature. Another factor that can significantly affect the oxidation of mercury across the SCR is the temperature of the flue gas. Thermodynamically, elemental mercury is more likely to be oxidized at lower temperatures, thus higher temperature applications present more of a challenge. In addition, if SO2 oxidation is a concern, catalyst chemistry, especially at higher temperatures, must be modified to help reduce such activity. This can adversely affect mercury oxidation performance, posing an additional consideration in the development of an advanced catalyst.
Reductants. As noted in Figure 2, oxidized mercury can be reduced back to its elemental form if there is an excess amount of reductants such as ammonia and SO2. Ammonia is the most significant reductant to be considered.
Catalyst Chemistry and Age. Catalyst chemistry is critical to driving mercury oxidation performance; however, it must be balanced with the other critical performance needs from the SCR, including NOx activity and SO2 oxidation. Understanding the interactive effects of the flue gas conditions and catalyst chemistry is critical to the optimization process. The age of the catalyst also affects the degree of mercury oxidation, as the catalyst potential decreases with time. The effects of catalyst aging on mercury oxidation can be minimized through a combination of proper chemical formulation and catalyst management procedures.
Other Glue Gas Constituents. The relative rate of mercury oxidation is higher in cases where the inlet concentration of Hg0 is higher. In a study conducted by the DOE, EPA, and Electric Power Research Institute, high levels of mercury oxidation over the SCR were observed at plants firing eastern bituminous coal where the level of Hg0 is high. However, oxidation was shown to be insignificant when most of the inlet mercury was already in the oxidized form.
Performance Modeling
Information gathered through a combination of significant laboratory testing and field data analysis over the past decade has resulted in the development of highly predictive performance models. These models provide the opportunity to prepare a parametric performance assessment to determine the best methods to achieve desired performance levels from the SCR. In order to set the desired performance target from the SCR, a full system evaluation must be considered. Typical questions that make up the model input include these:
- What is the overall system goal?
- What are the fuels and fuel blends to be considered?
- What are the operating conditions and other key performance requirements of the SCR, such as NOx reduction and SO2 conversion?
- What level of mercury oxidation should be expected through the air preheater?
- Will the dust collection system contribute to mercury reduction performance?
- Will fuel or postcombustion additives be utilized?
- What collection efficiency should be expected from the FGD system, including consideration of re-emission control (conversion back to elemental mercury)?
- What are the economic considerations, including, but not limited to, cost of additives, cost of catalyst, pressure loss, impact on ash sales, boiler corrosion, and material-handling equipment?
Once requirements are understood, a series of “what-if” scenarios can be run and supplemented with additional testing as needed. The net result will allow the user to assess the best method(s) to achieve the desired performance requirements at the lowest total ownership cost.
Catalyst Advancements for Increased Mercury Oxidation
We have presented many of the factors that affect the rate and amount of mercury oxidation in an SCR catalyst. As discussed, two of the larger influences are halogen content and temperature.
Mitsubishi Heavy Industries and Cormetech have developed an advanced catalyst tailored to oxidize mercury to high levels under challenging conditions involving high temperatures and low HCl concentrations, both of which negatively impact mercury oxidation across conventional SCR catalysts.
Let’s now examine the performance differences between advanced Hg oxidation SCR catalysts and standard SCR catalyst products with respect to halogen content and temperatures.
Aged Catalyst Performance. Figure 3 shows an example of actual field performance versus predictive models for an aged SCR catalyst at varying levels of HCl in the flue gas. At a temperature of approximately 690F, the percentage of oxidized mercury at the SCR outlet measured during actual tests on aged catalyst ranged from 75% to 90% over a range in flue gas HCl concentration of 10 to 25 ppmvd. The predicted levels of oxidized mercury increase to >95% levels at higher levels of HCl.
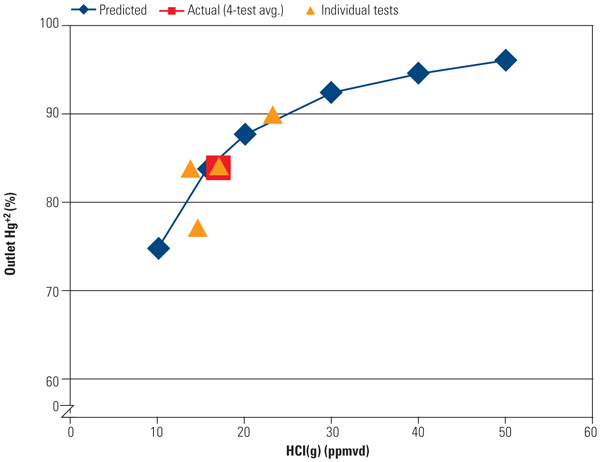 |
| 3. Mercury rising. Oxidized mercury levels increase with rising HCl concentrations in the flue gas. Data were taken from a large utility boiler operating at full load with 12,000 operating hours on the SCR. Source: Cormetech Inc. |
Advanced Catalyst Performance. Figure 4 illustrates the performance of the advanced Hg oxidation catalyst versus standard catalyst at extremely low HCl concentrations and moderate flue gas temperatures (5 ppmvd and 700F). The advanced catalyst exhibited high levels of oxidized mercury for both fresh and aged catalyst and shows a substantial increase over an existing standard catalyst.
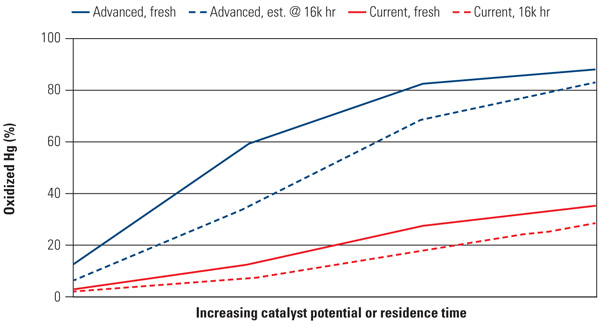 |
| 4. Low HCl operation. Advanced SCR catalysts demonstrate better mercury oxidizing levels over time. Data were taken at 5 ppm HCl and 700F. Source: Cormetech Inc. |
In Figure 5, the percentage of oxidized mercury is shown, as expected, to be greater due to the higher HCl concentration, despite a higher temperature (65 ppmvd and 757F).
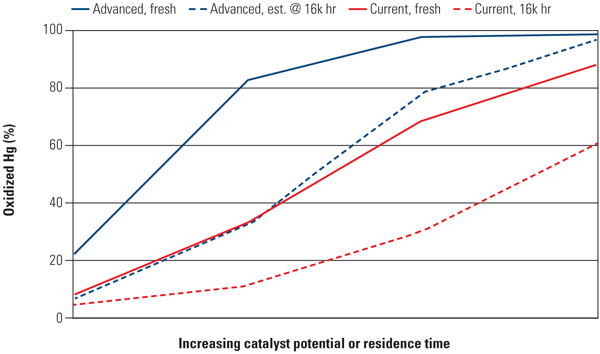 |
| 5. High HCl operation. Advanced SCR catalysts, at high levels of HCl (65 ppm) in the flue gas, also exhibit improved mercury oxidizing potential over time. Data were taken at 757F. Source: Cormetech Inc. |
Planning for the Future
It’s reasonable to assume that the performance of mercury reduction technologies and other co-benefits, such as we’ve discussed here, will be used to set future maximum achieveable control technology regulations. Regardless of future limits, we suggest that you adopt a holistic approach for optimizing mercury reduction that considers all the components in your air quality control system, from the coal pile through the stack. Only then will you meet your goal of maximizing mercury removal while minimizing overall system and operating costs.
—Scot Pritchard (pritchardsg@ cormetech.com) is vice president, sales & marketing for Cormetech Inc. Masashi Kiyosawa (masashi_ [email protected]) is manager, environmental equipment team, Mitsubishi Heavy Industries, Nagasaki, Japan. Katsumi Nochi ([email protected]) is senior researcher, Chemical Process Laboratory, Mitsubishi Heavy Industries, Hiroshima, Japan.